🎯 Big picture
You’re given a substrate, a nucleophile or base, and maybe even a solvent. Your task is to predict which reaction pathway it will take: substitution (SN1 or SN2) or elimination (E1 or E2).
To figure that out, we evaluate three main factors: substrate structure, nucleophile or base strength, and temperature. These factors help guide you toward the correct mechanism.
Solvent choice is important too—not because it changes the mechanism, but because it can make a reaction dramatically faster or slower.
We’ll begin with a summary table to give you a quick overview, then dive into each step with detailed reasoning and examples.

Step 1: Classify the substrate
The substrate is the molecule that holds the leaving group. Most commonly, you’ll see alkyl halides or sulfonates like tosylates.
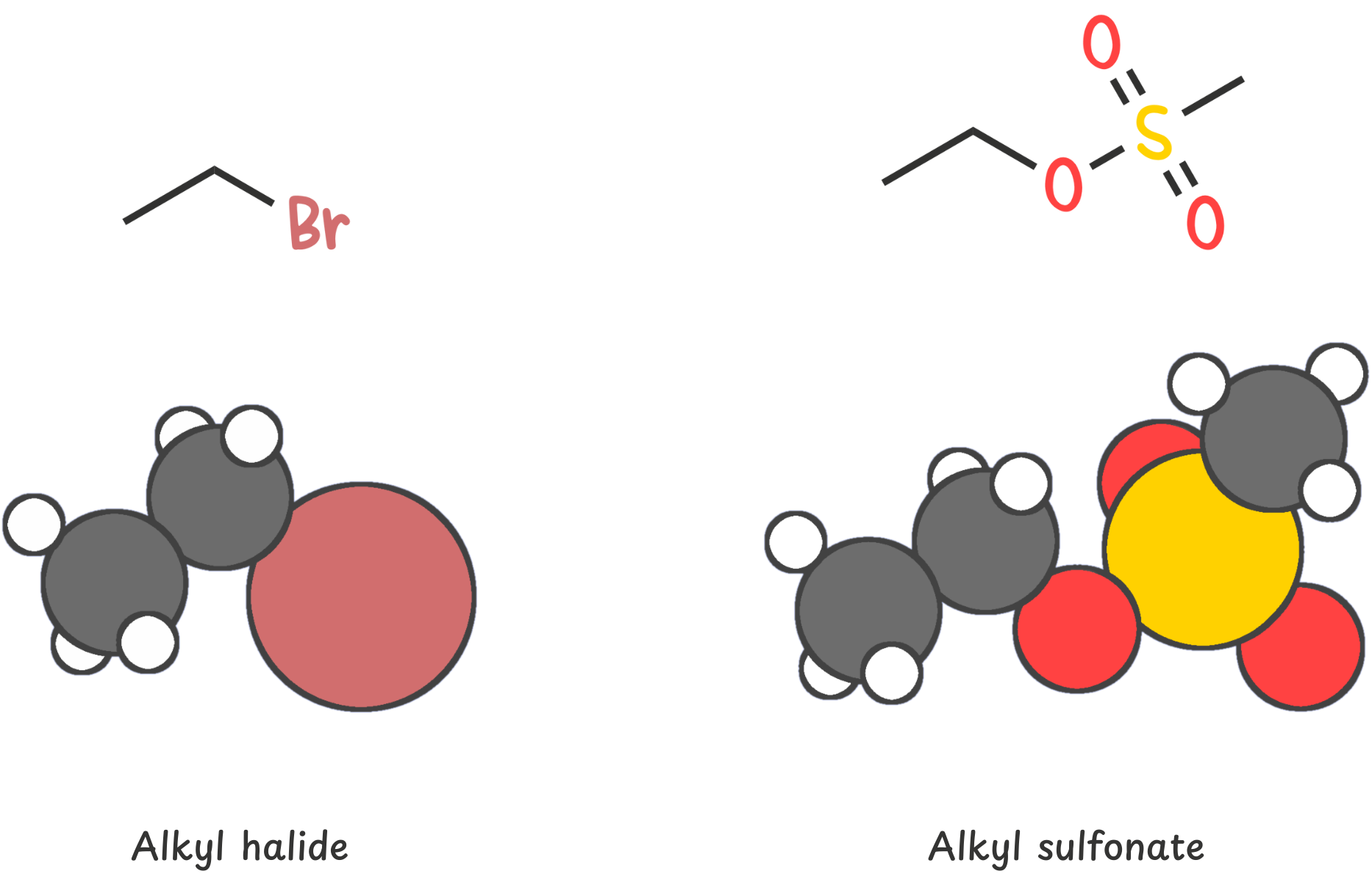
Start by identifying the carbon directly bonded to the leaving group. This is the electrophilic center.
Ask yourself: How substituted is that carbon? In other words, how many other carbon groups are attached to it?
Let’s unpack that.
Methyl (CH3X)
Only SN2 reactions are possible. Elimination cannot occur because methyl substrates lack β-hydrogens, and substitution through SN1 is ruled out since methyl cations are far too unstable to form. As a result, both SN1 and E1 pathways are effectively impossible.
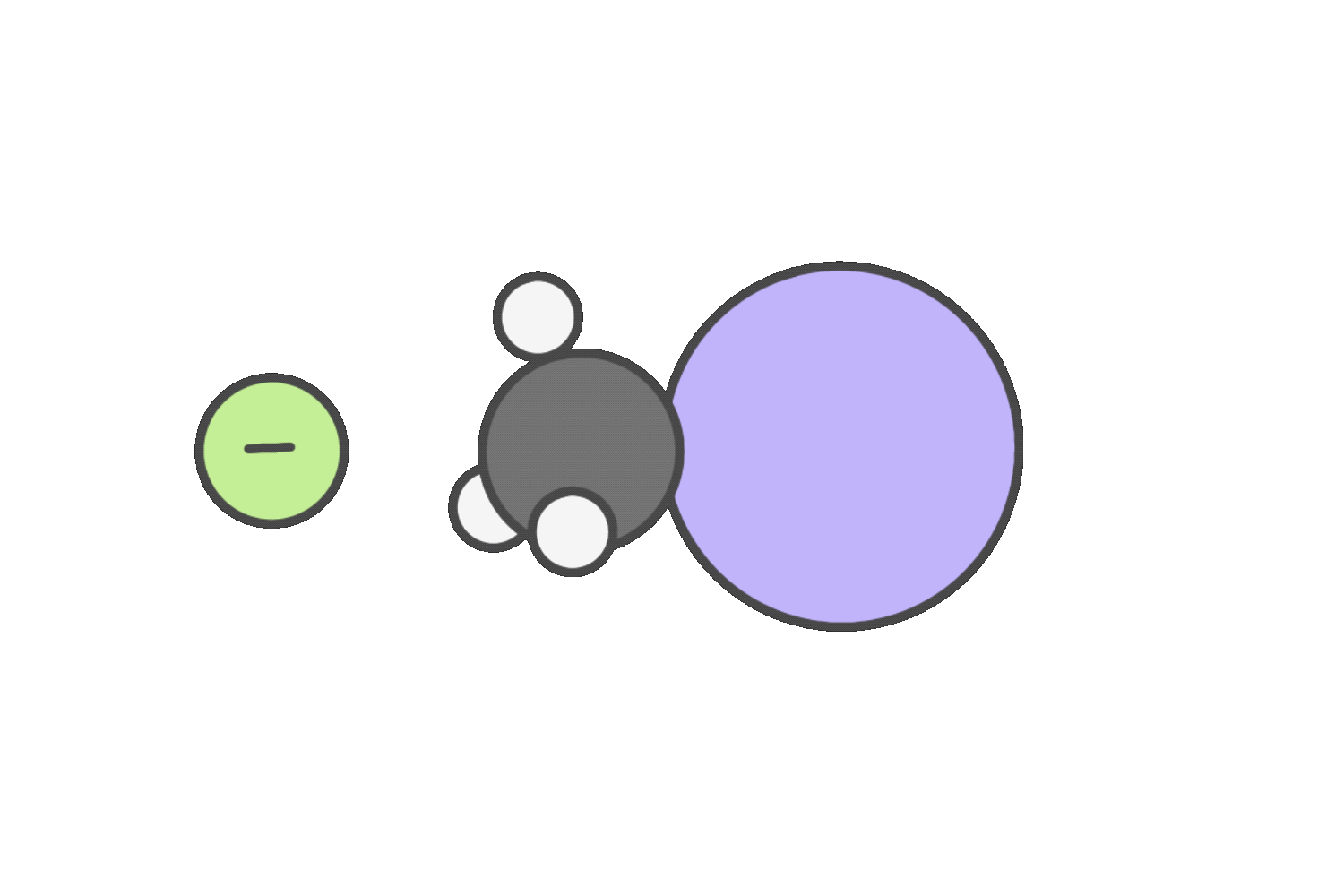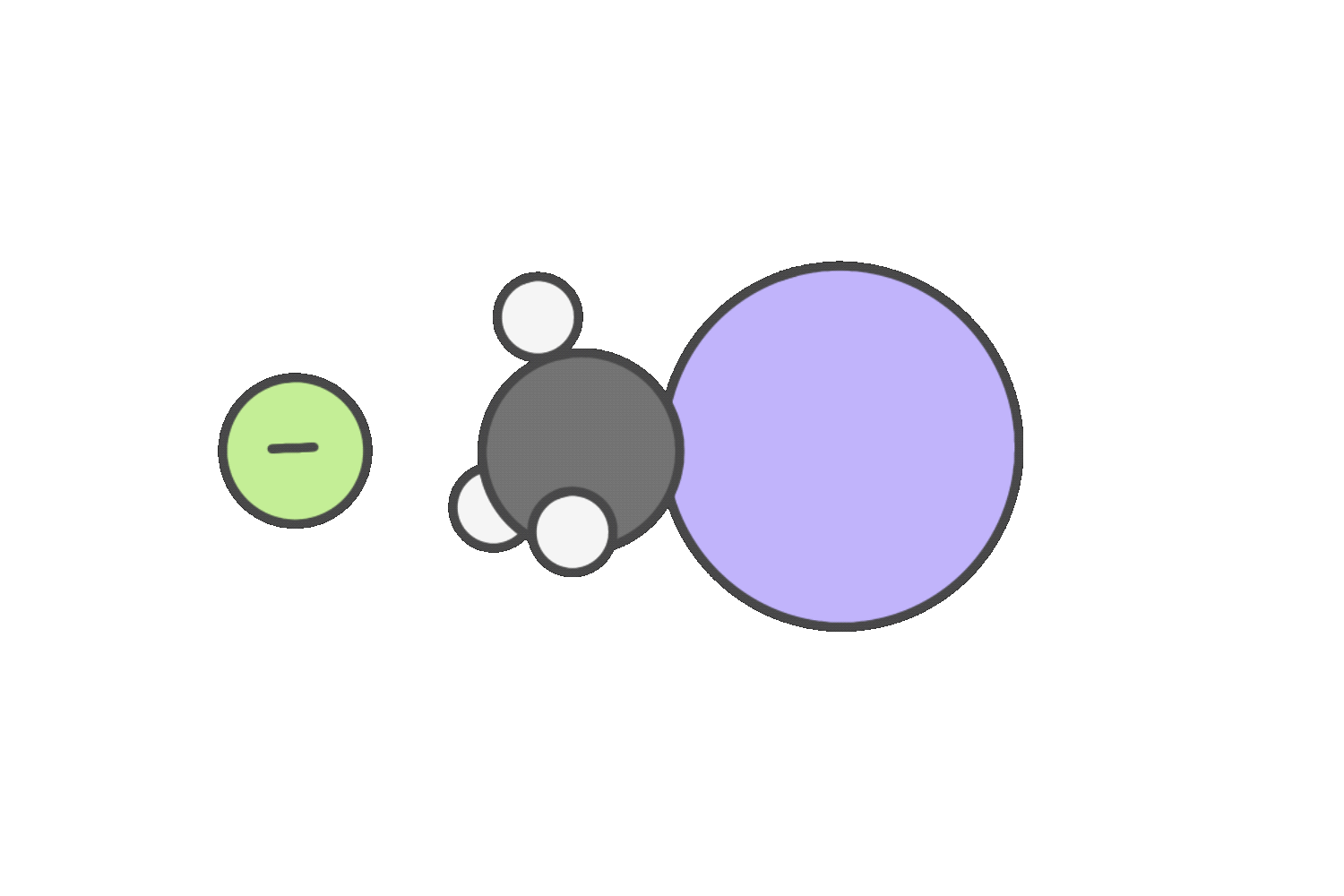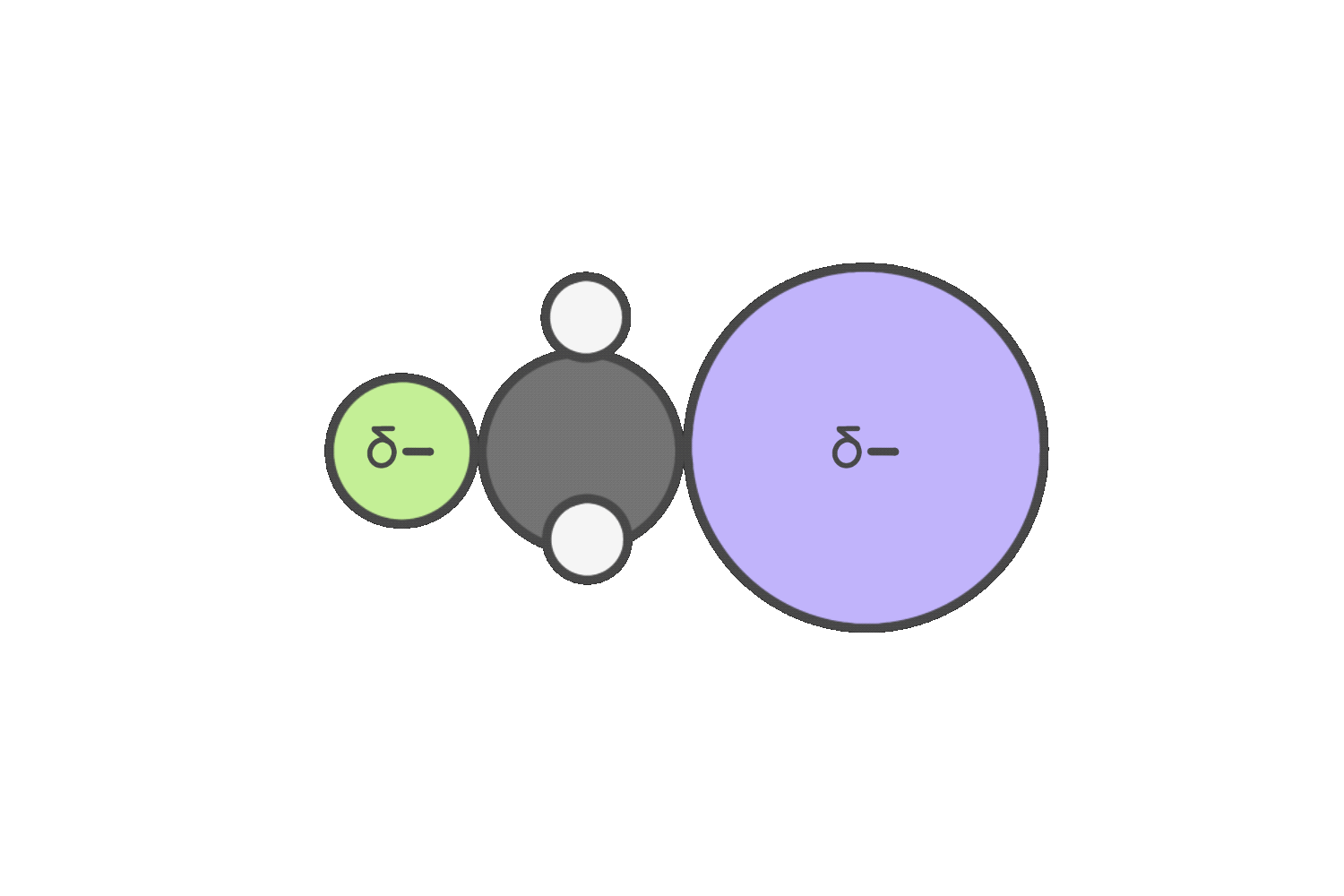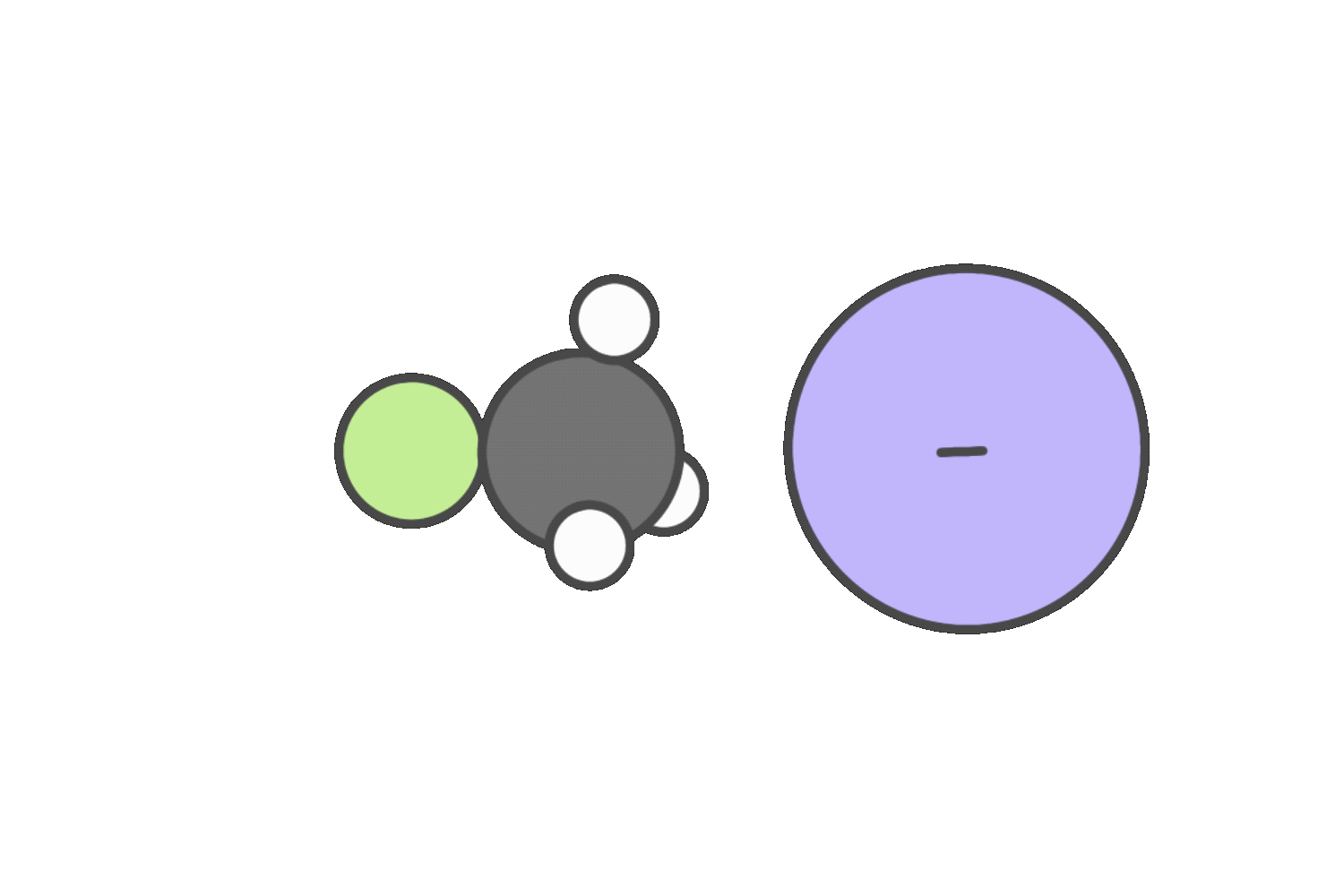

Primary
SN2 is favored when you have a strong nucleophile that’s also a weak base, such as iodide or azide.
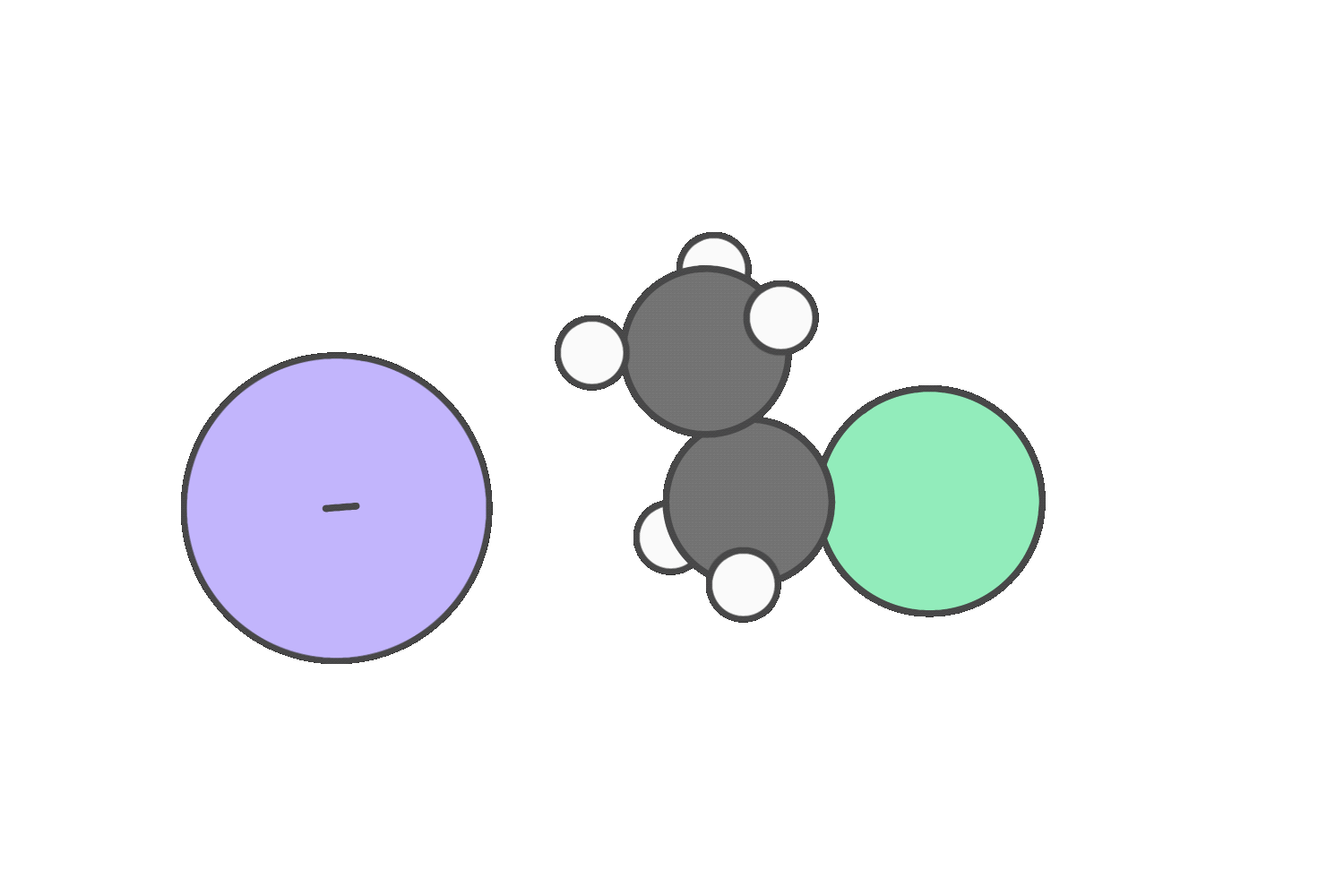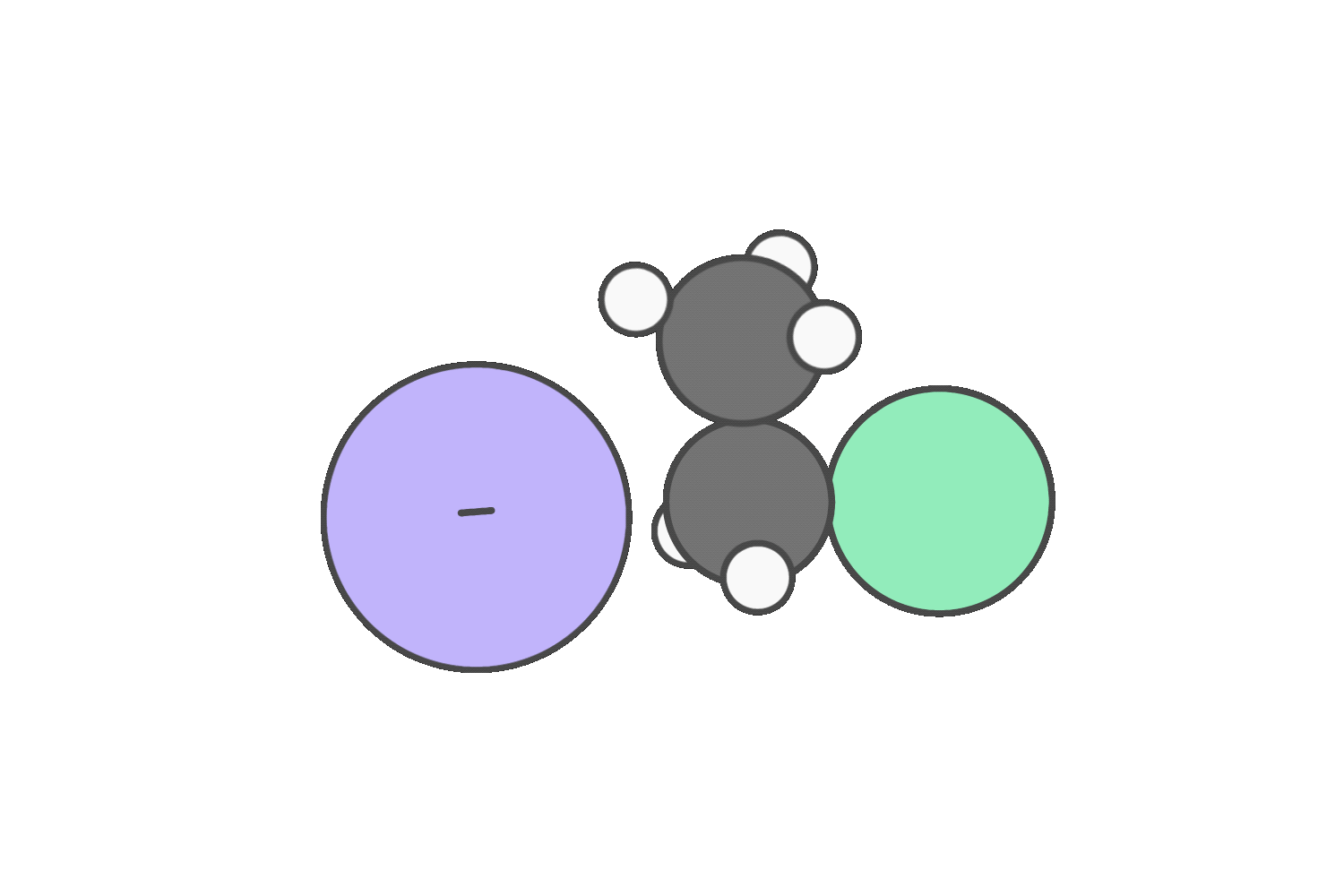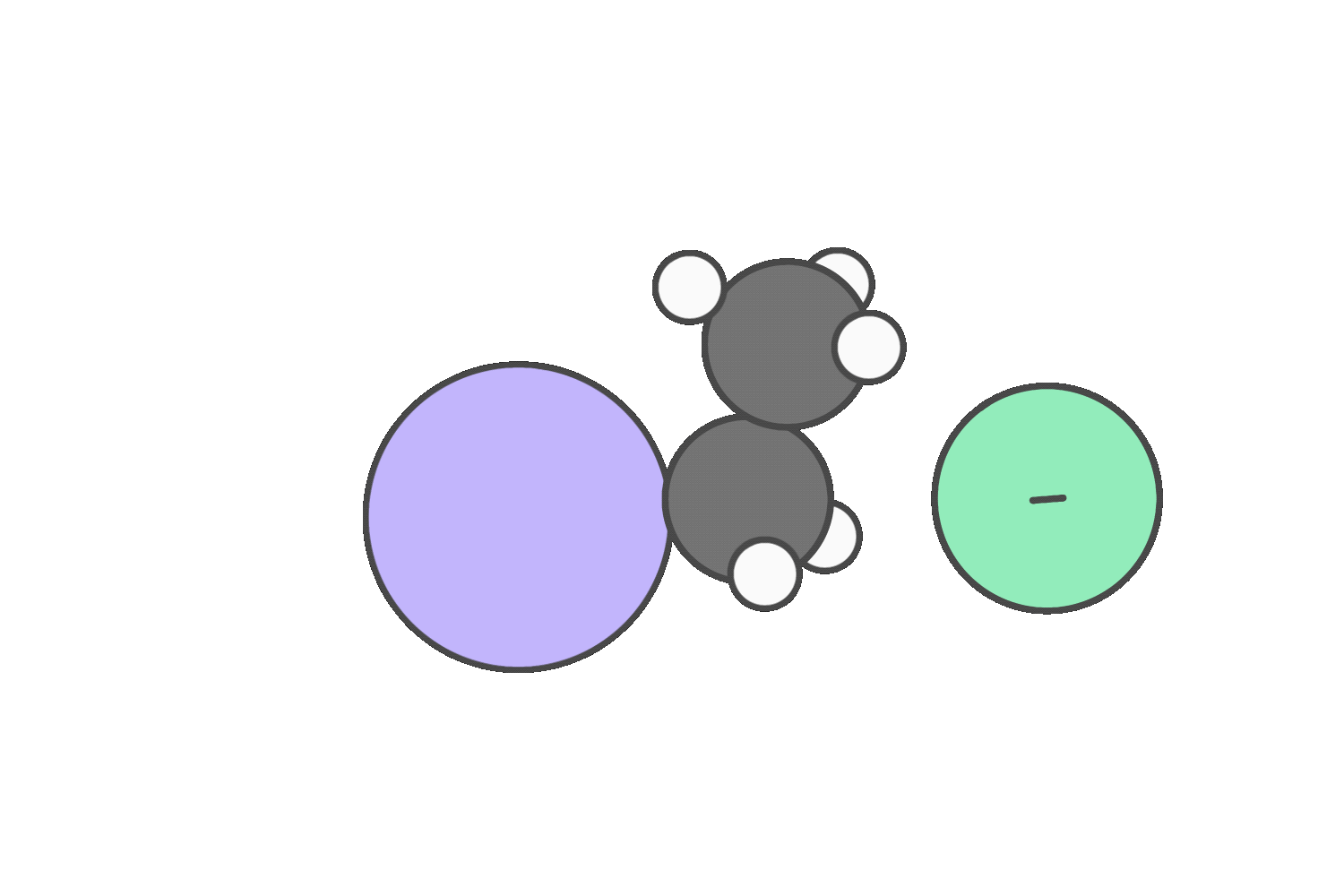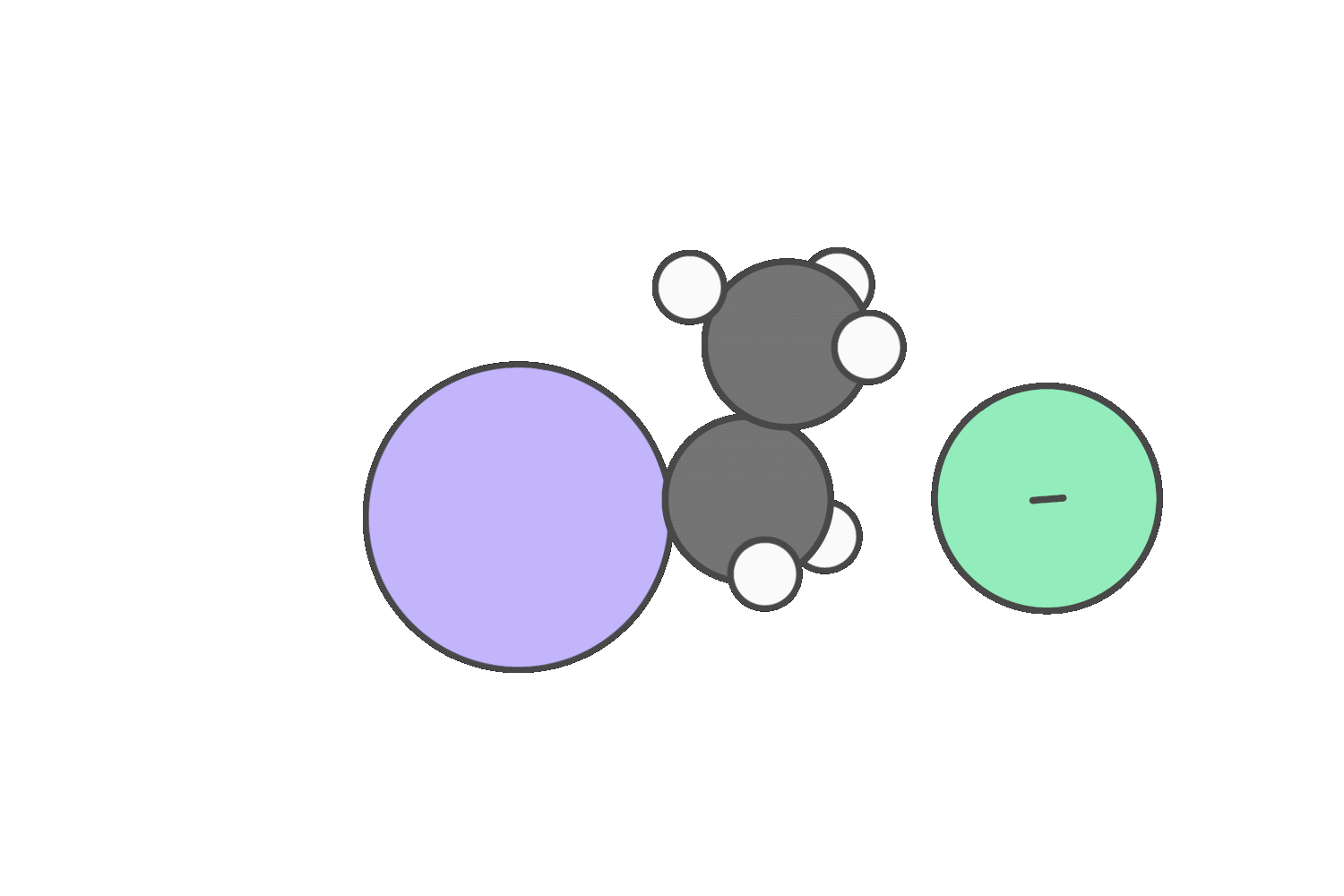

But if the reagent is a strong base that’s a poor nucleophile (like tert-butoxide), steric hindrance blocks backside attack, making E2 more favorable.
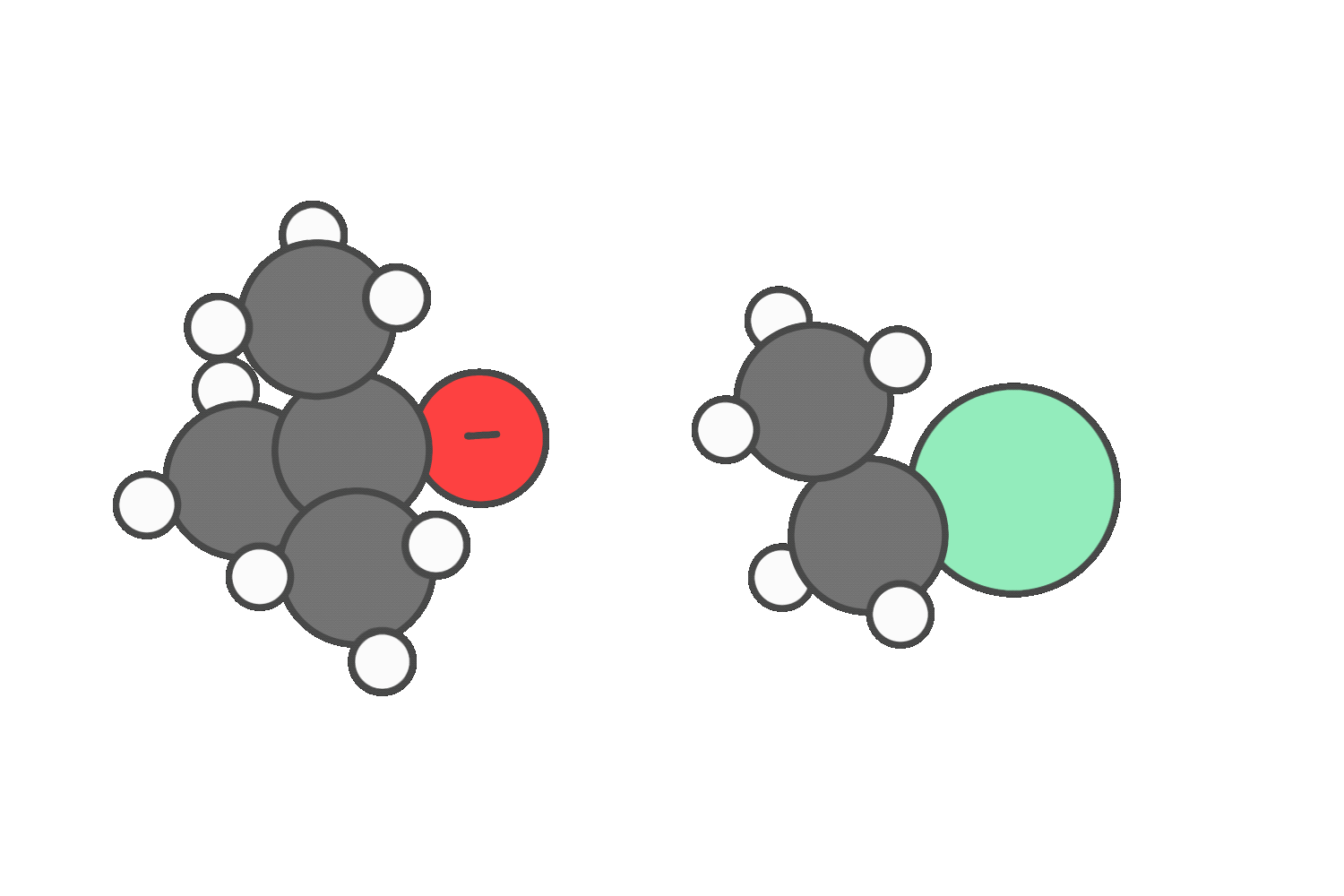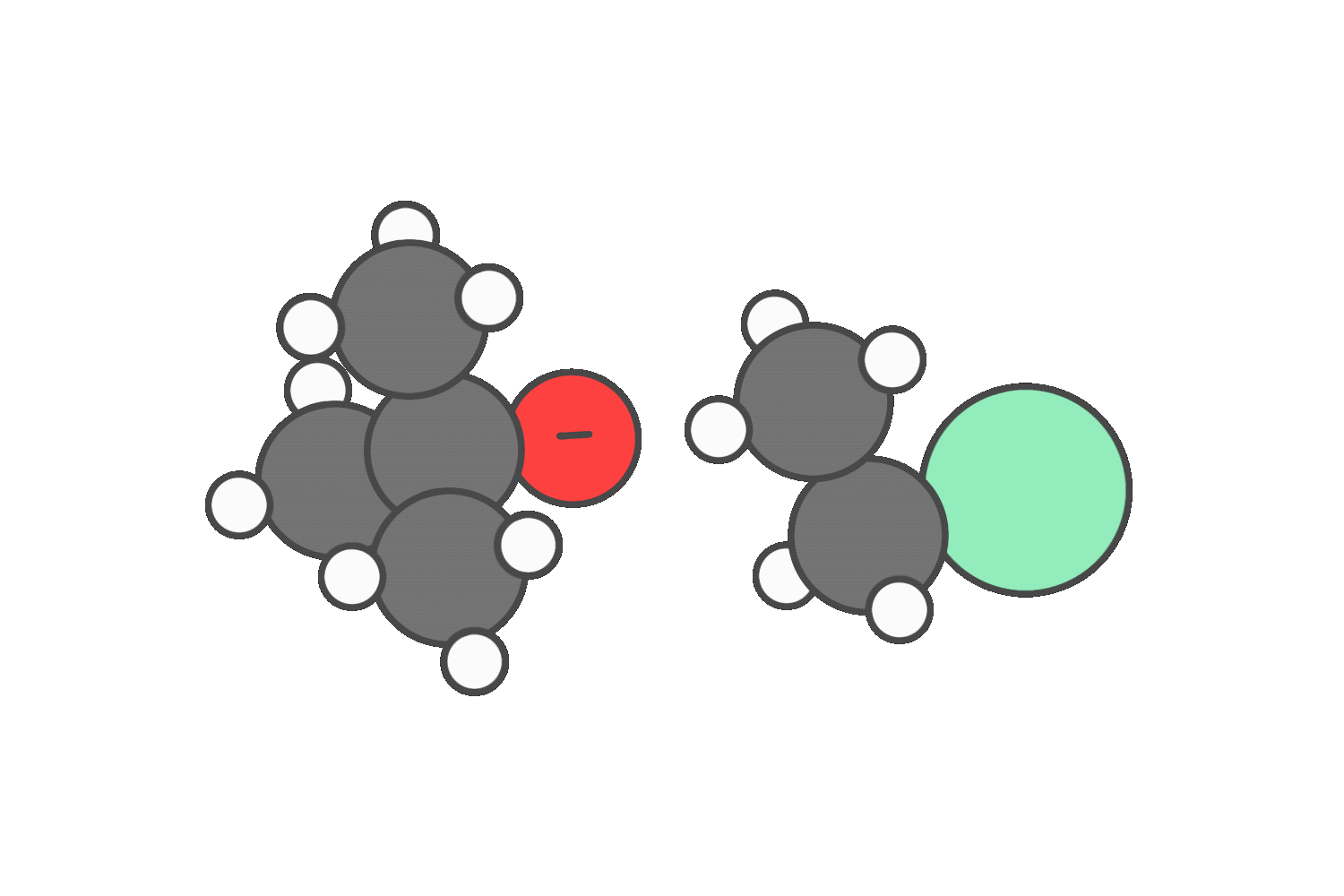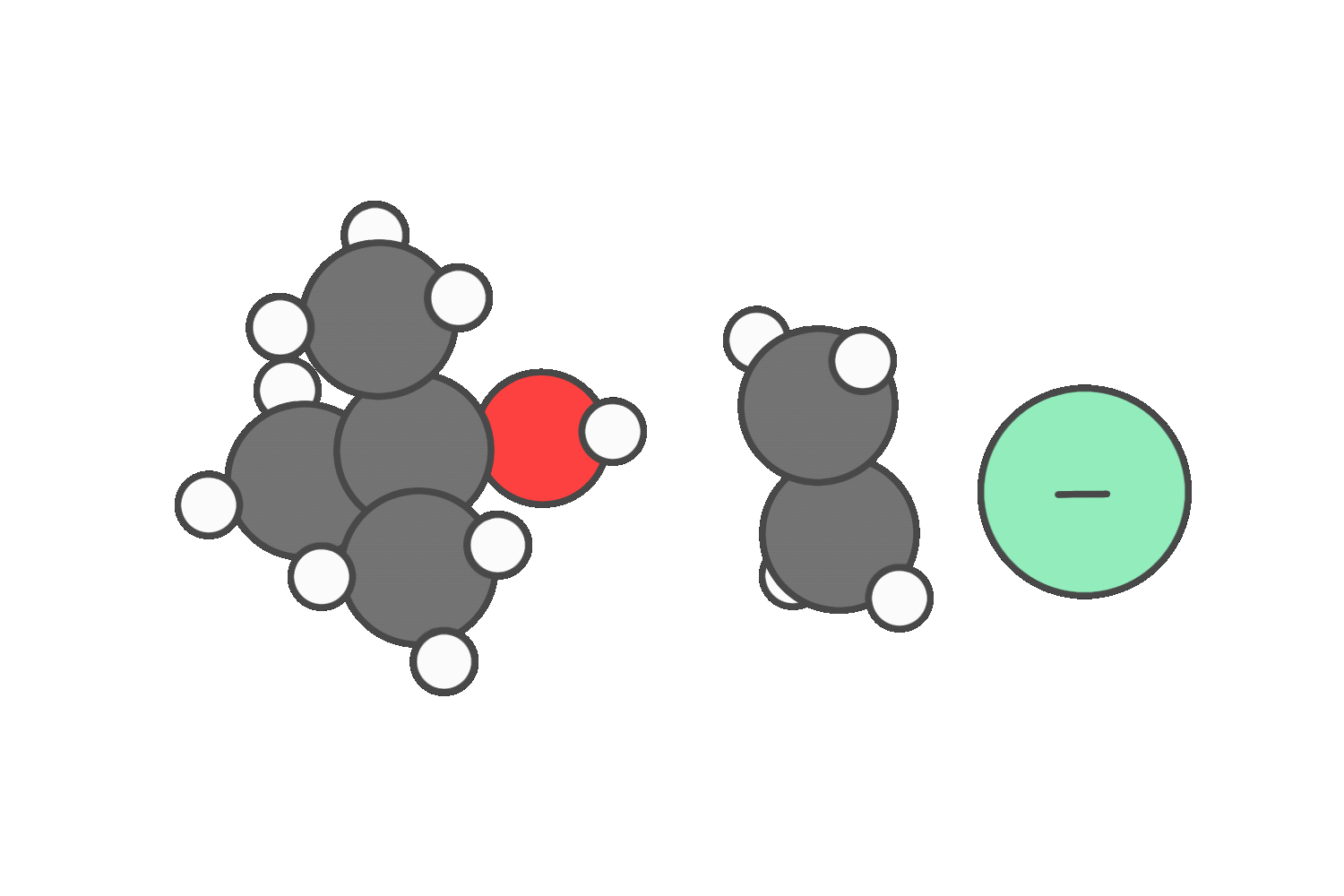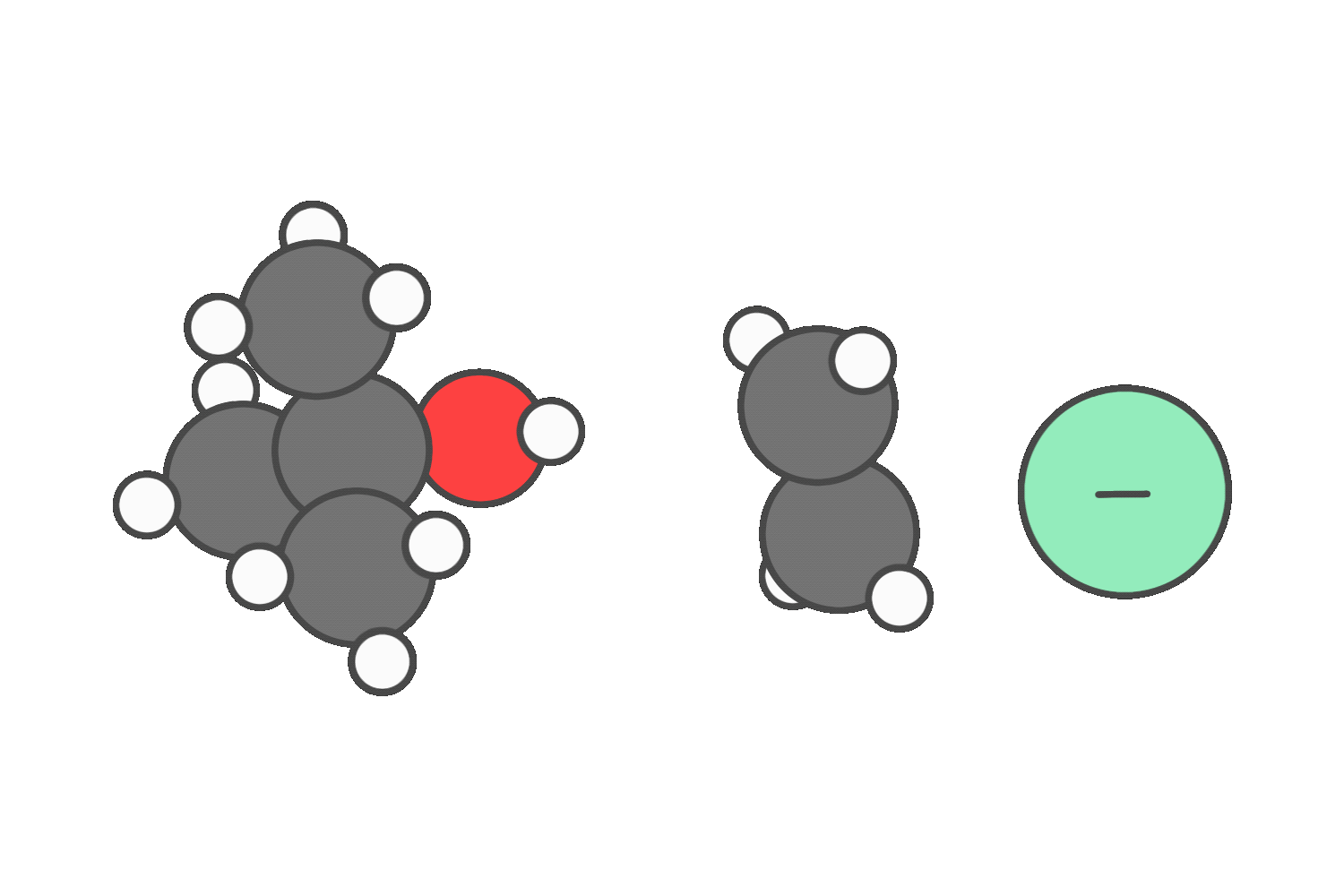
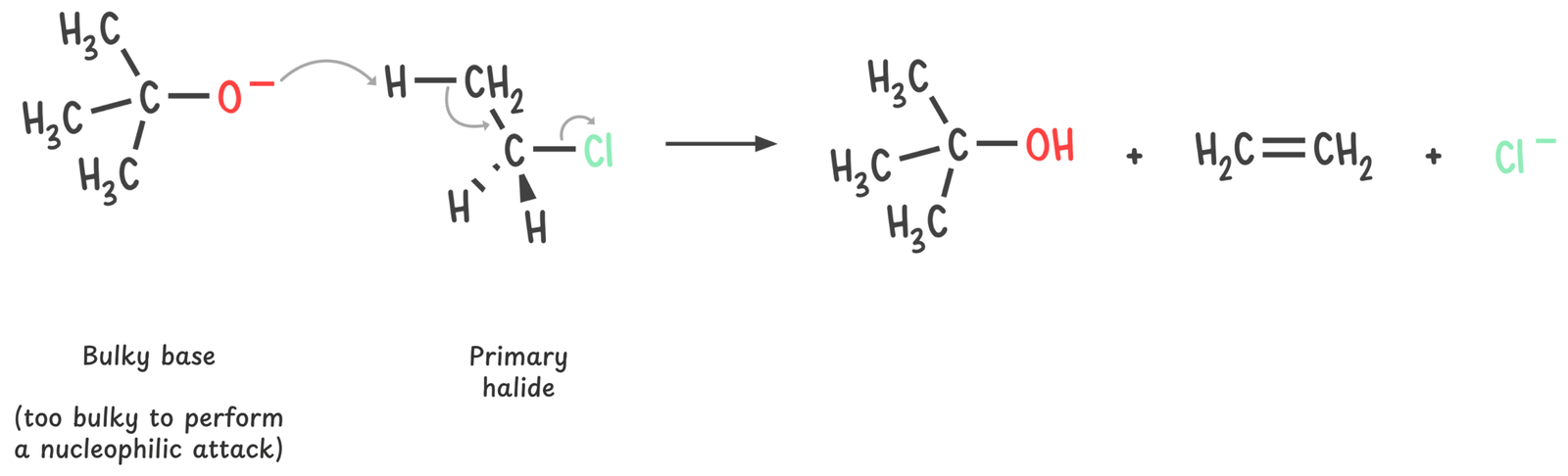
When the reagent is both a strong nucleophile and a strong base (such as hydroxide or ethoxide), both SN2 and E2 are possible.
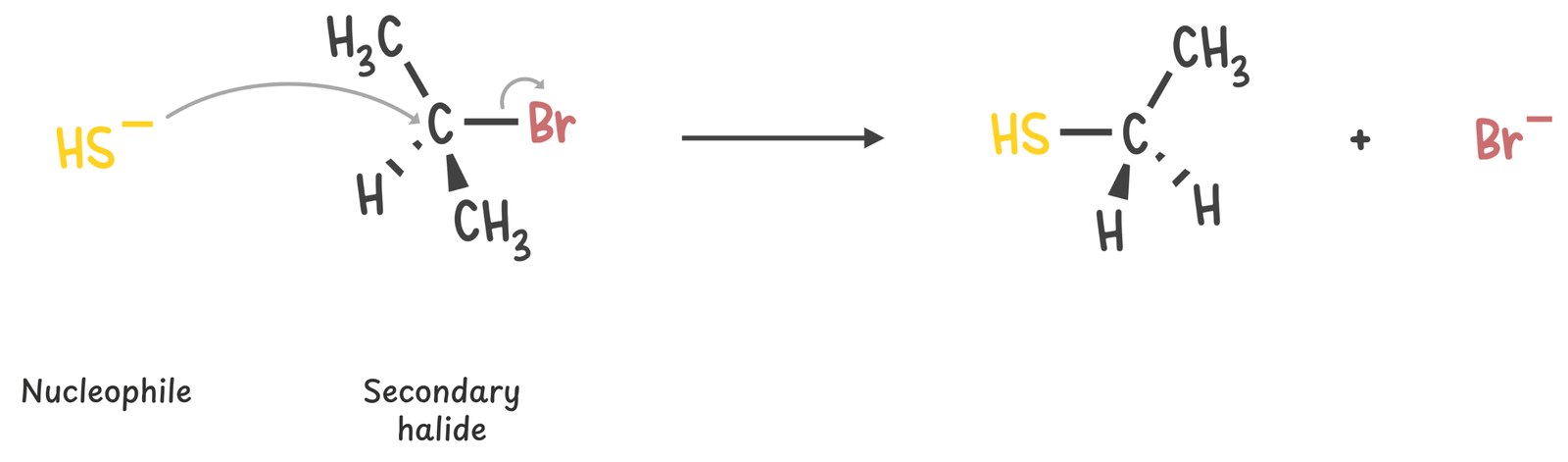
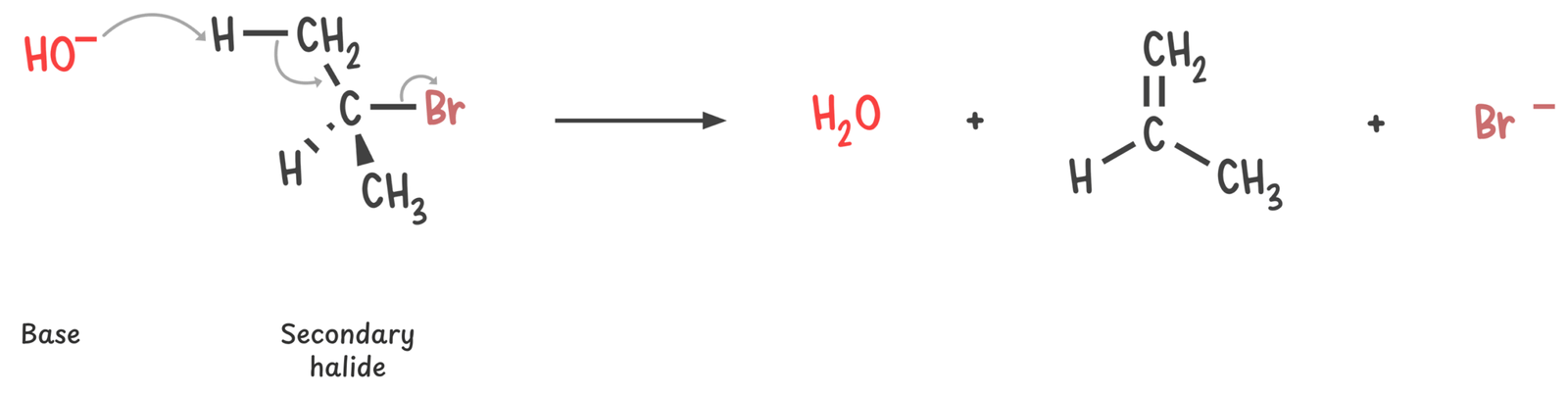
Secondary
This is the trickiest class. These substrates can react through SN1, SN2, E1, or E2 depending on the other conditions.
Strong nucleophiles that are weak bases tend to favor the SN2 pathway.
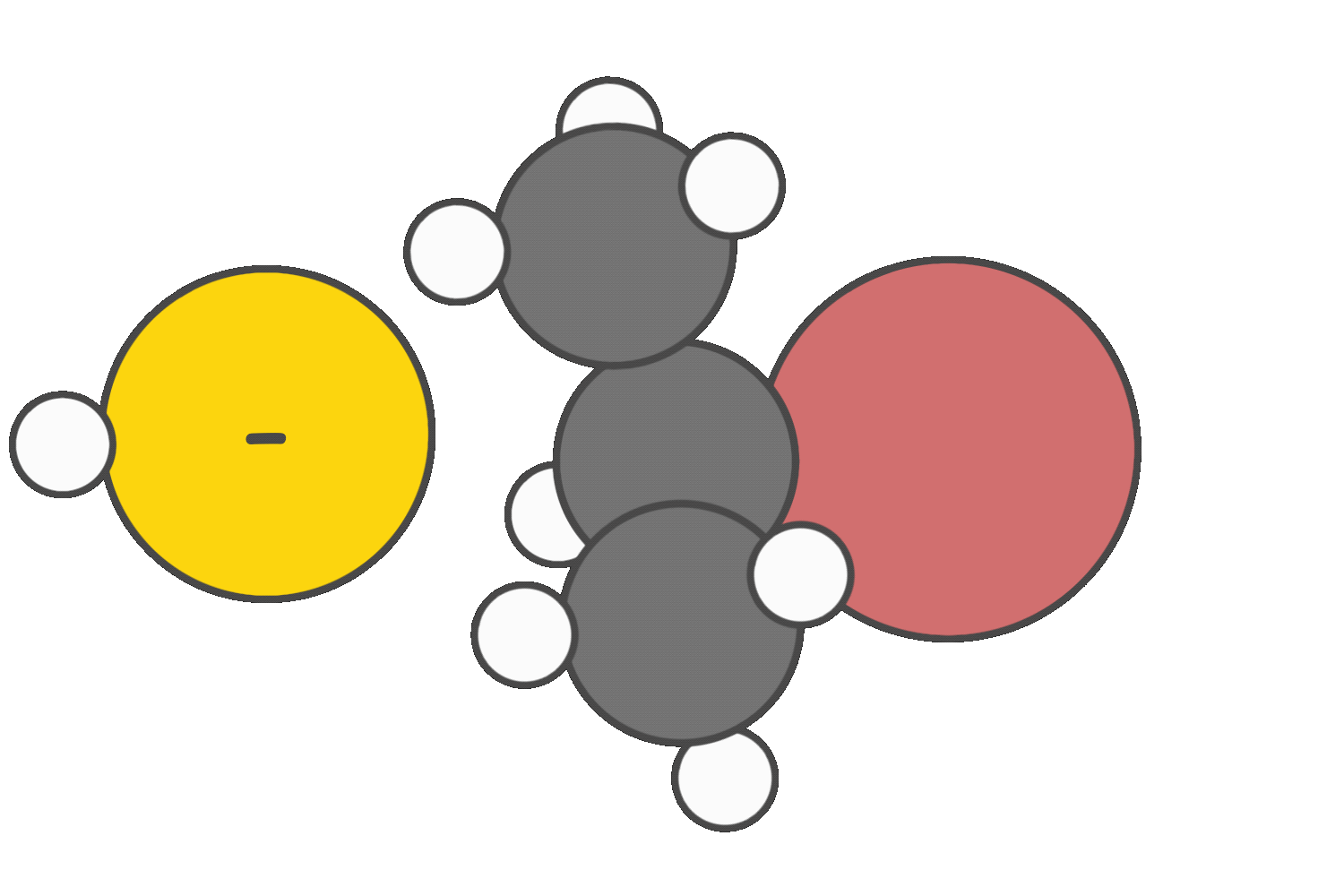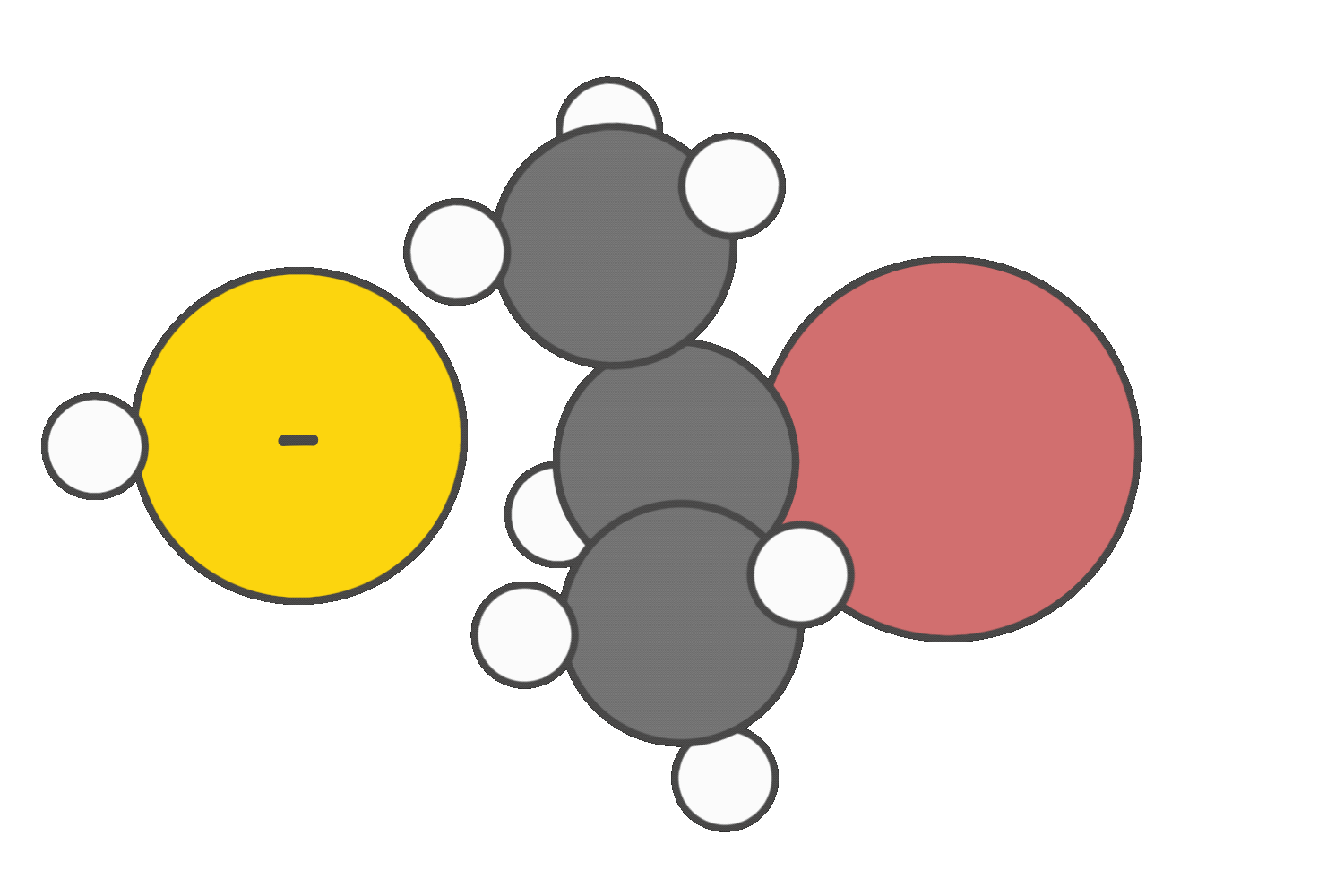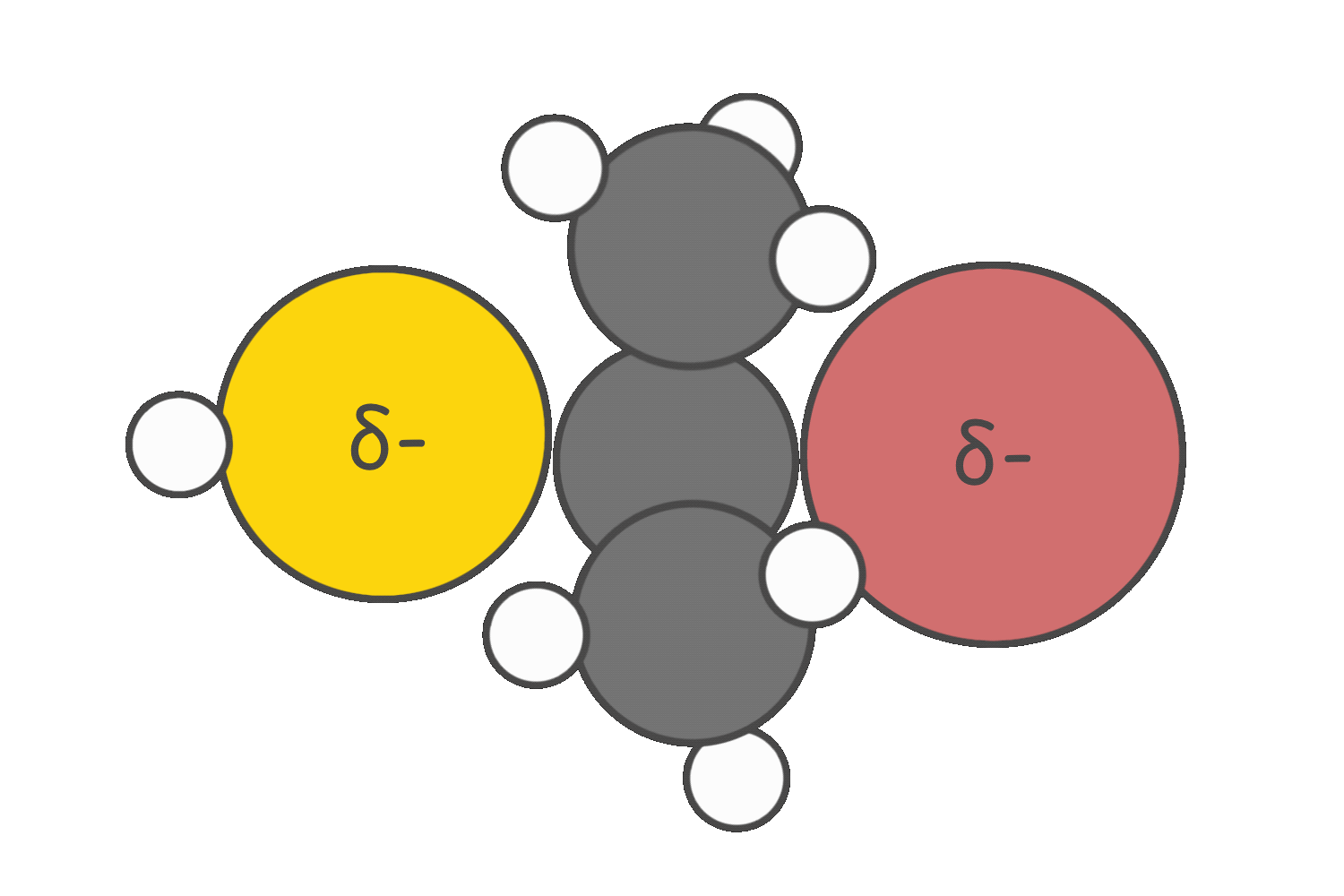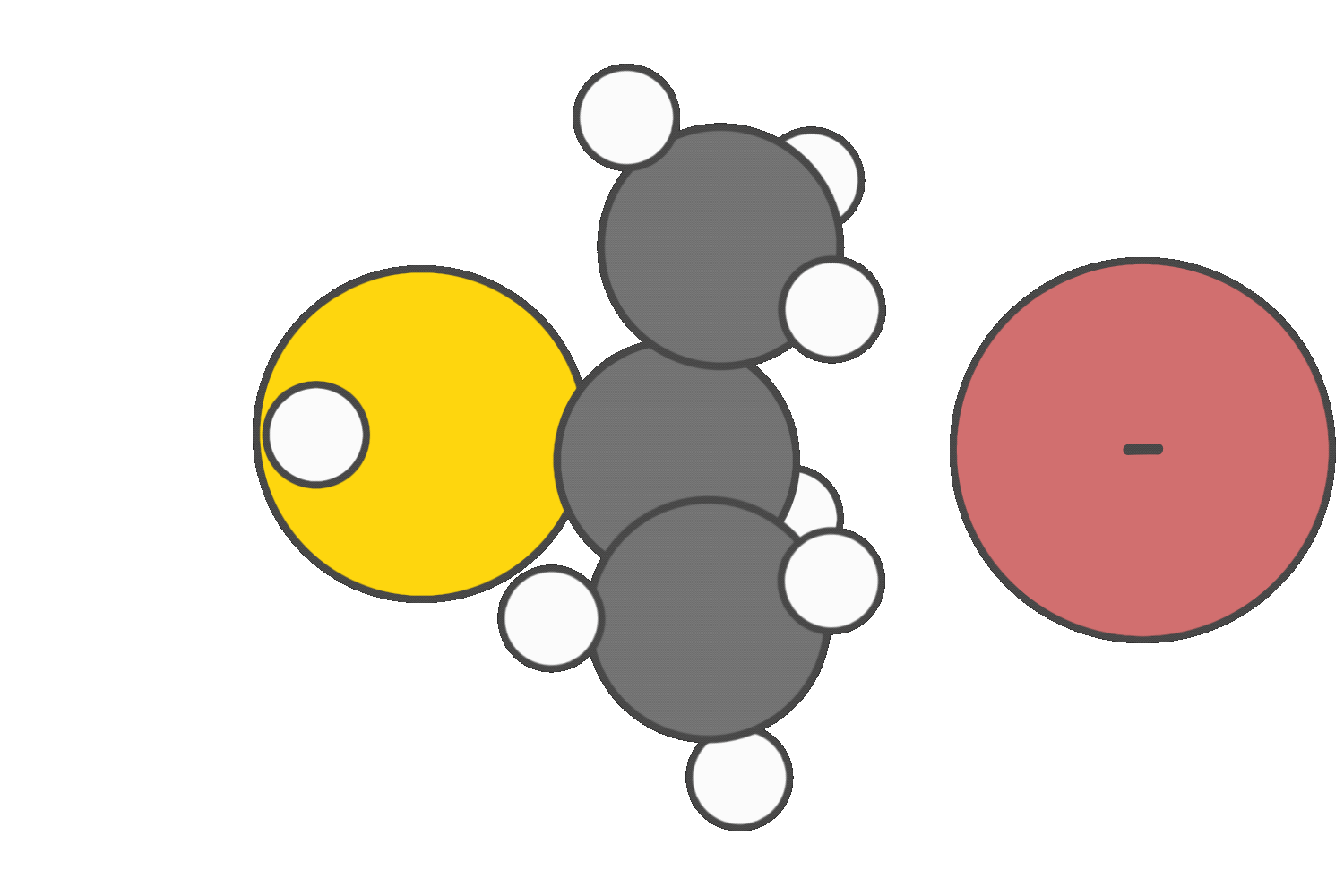
Strong bases promote E2 elimination.
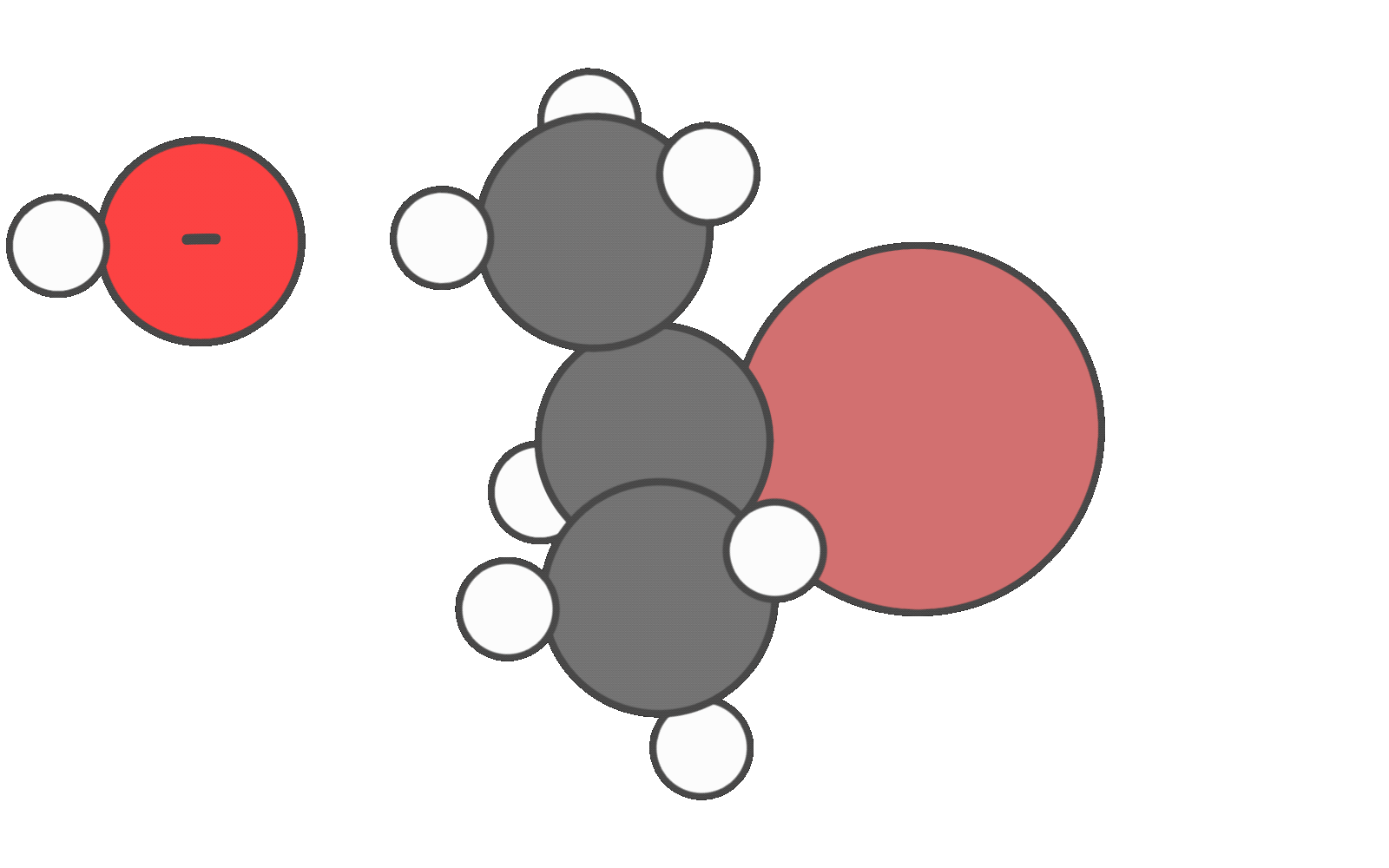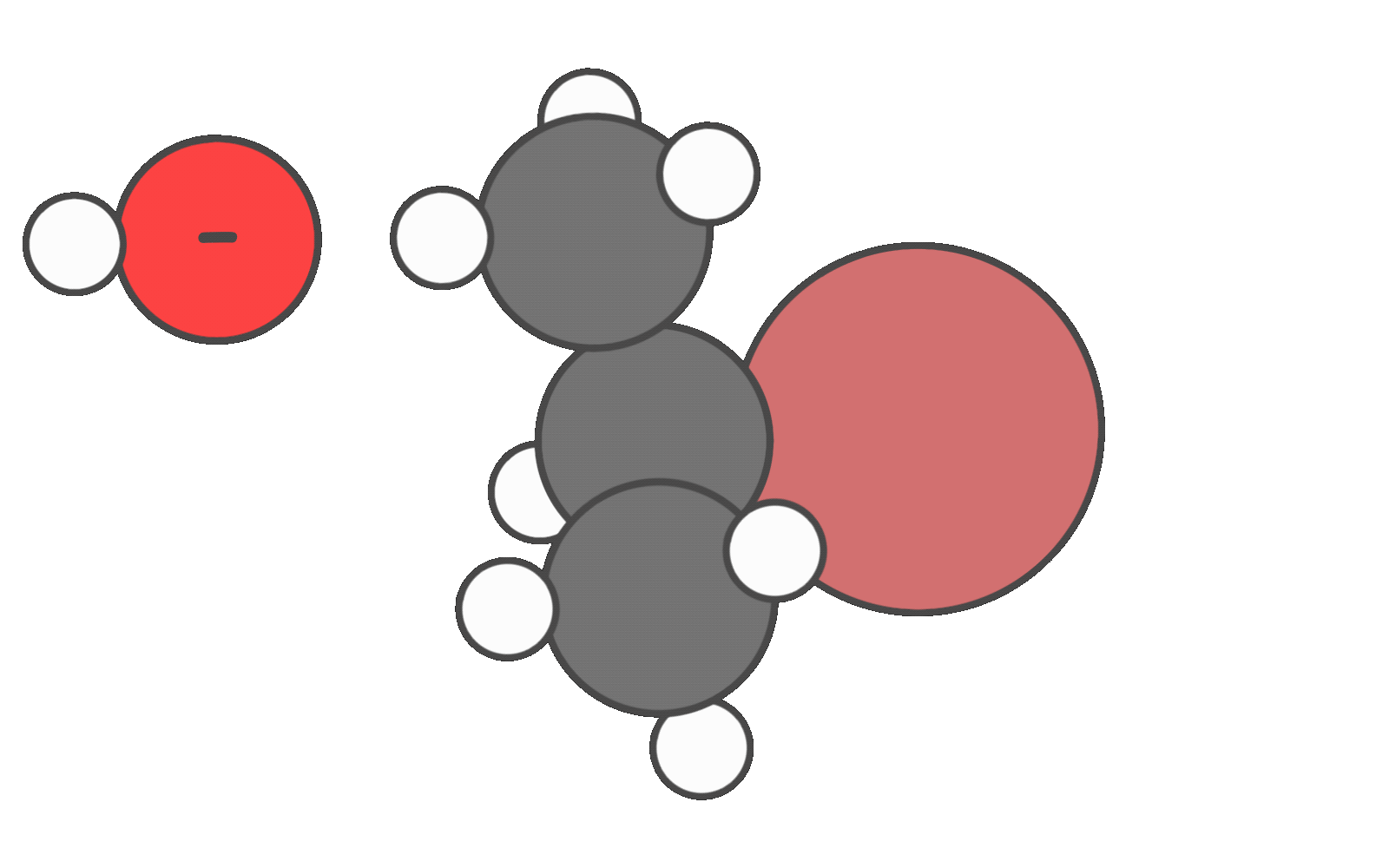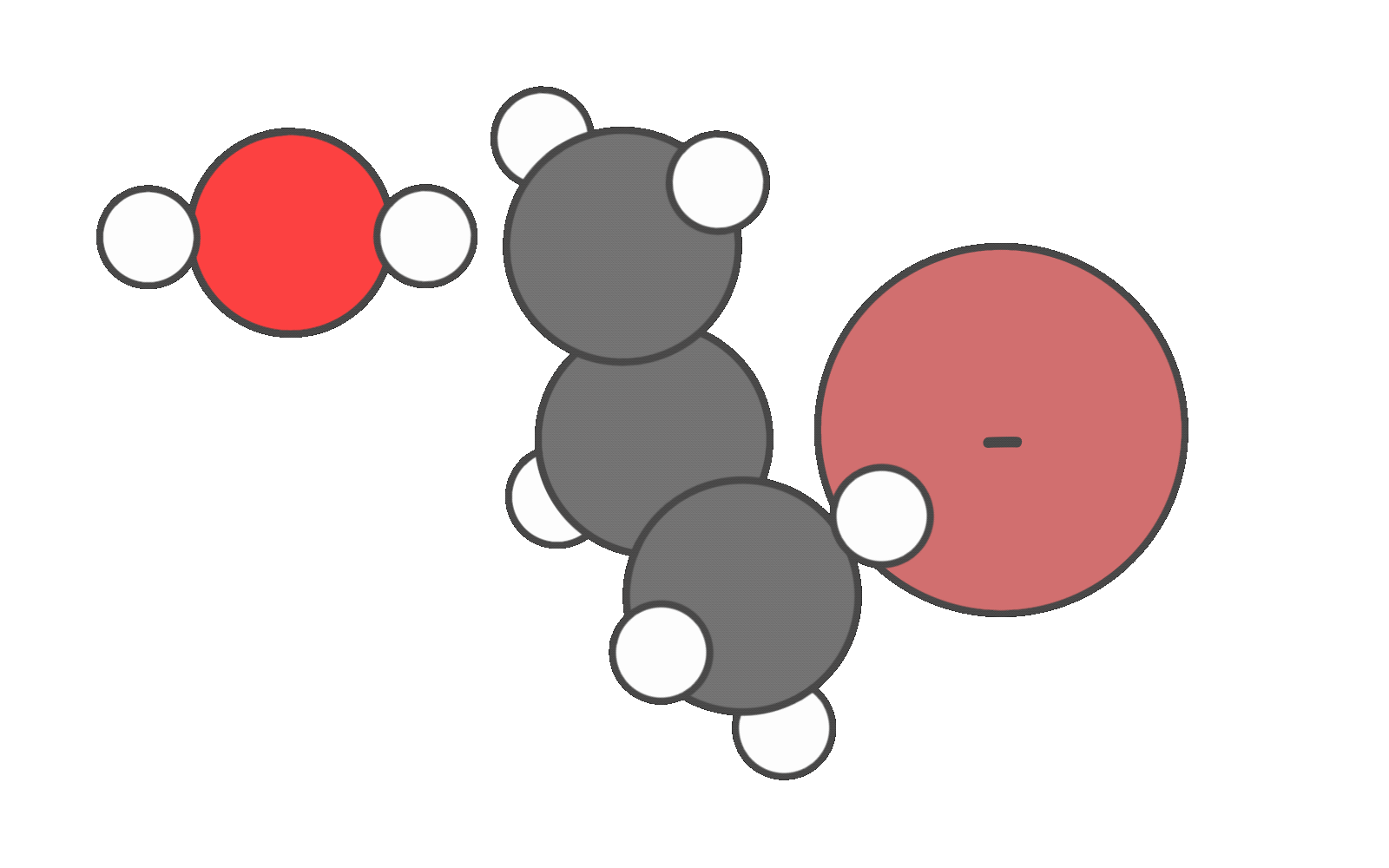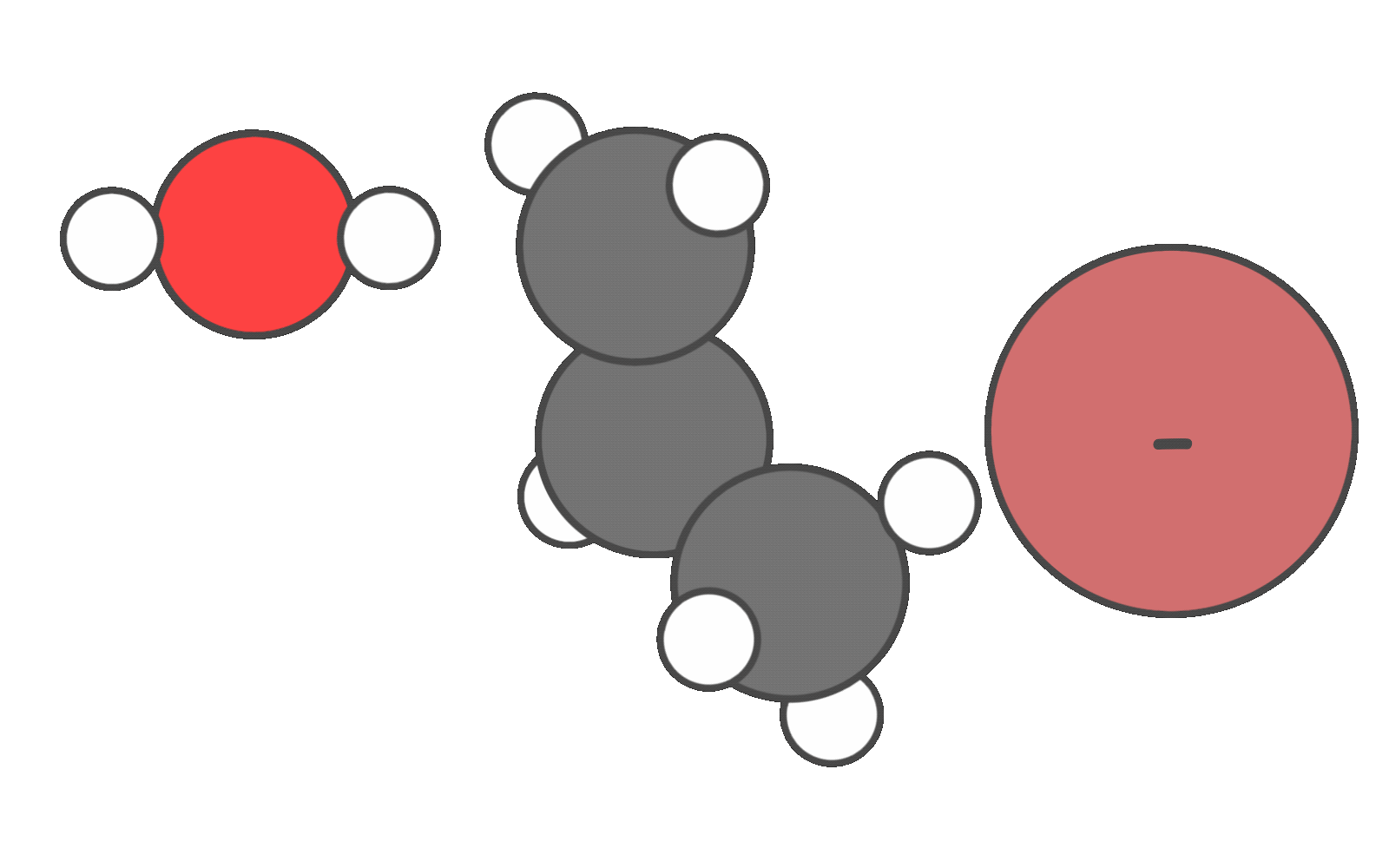
When weak nucleophiles that are weak bases are used in polar protic solvents, the reaction may proceed through the SN1/E1 mechanism.

However, secondary substrates are generally poor candidates for SN1/E1, and SN2 due to limited carbocation stability and moderate steric hindrance. As a result, E2 is often the most favorable pathway, especially in the presence of a strong base.
Tertiary
Tertiary substrates are too sterically hindered to allow backside attack, so they do not undergo SN2 reactions. Substitution, if it occurs, must proceed through the SN1 mechanism, which involves carbocation formation.
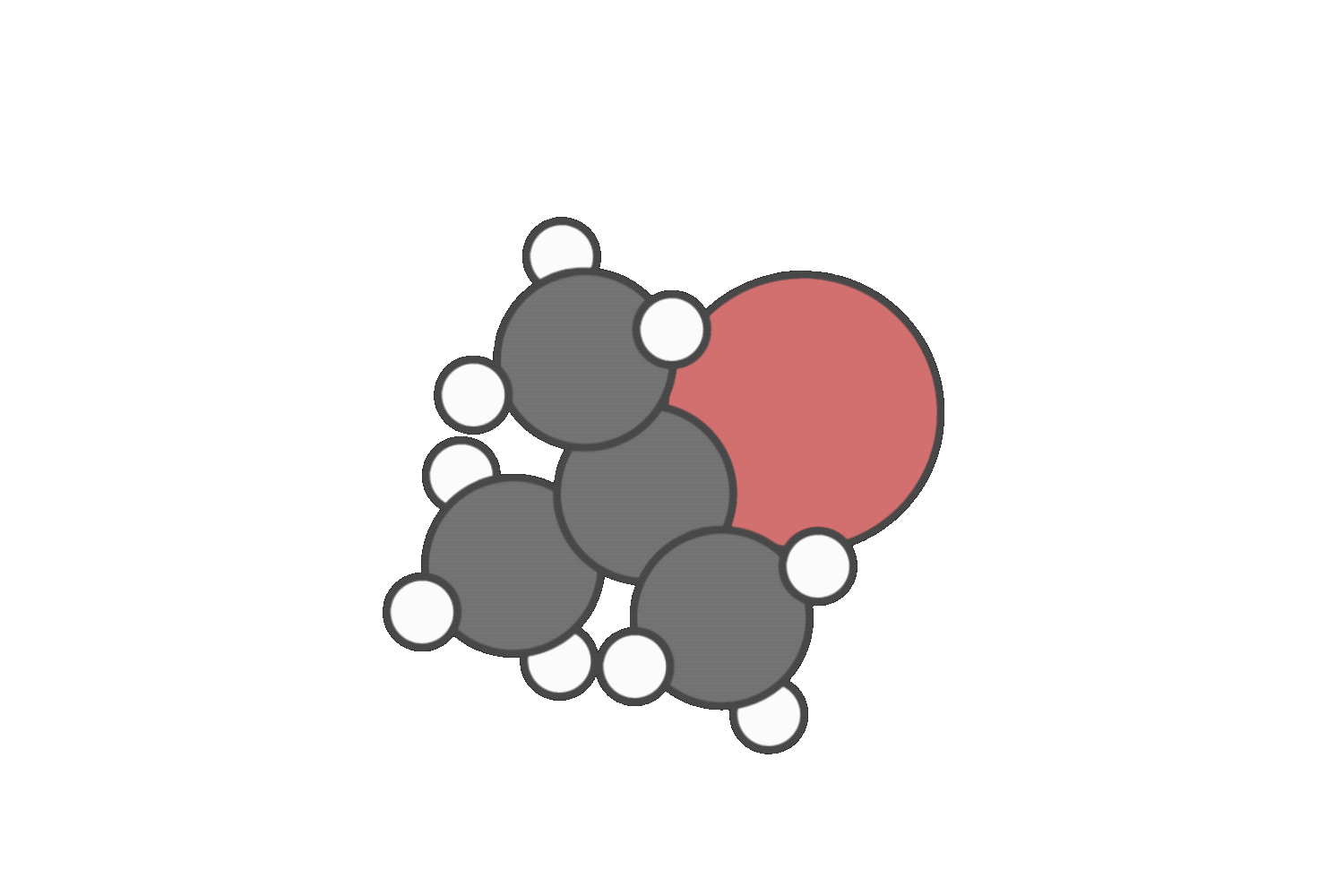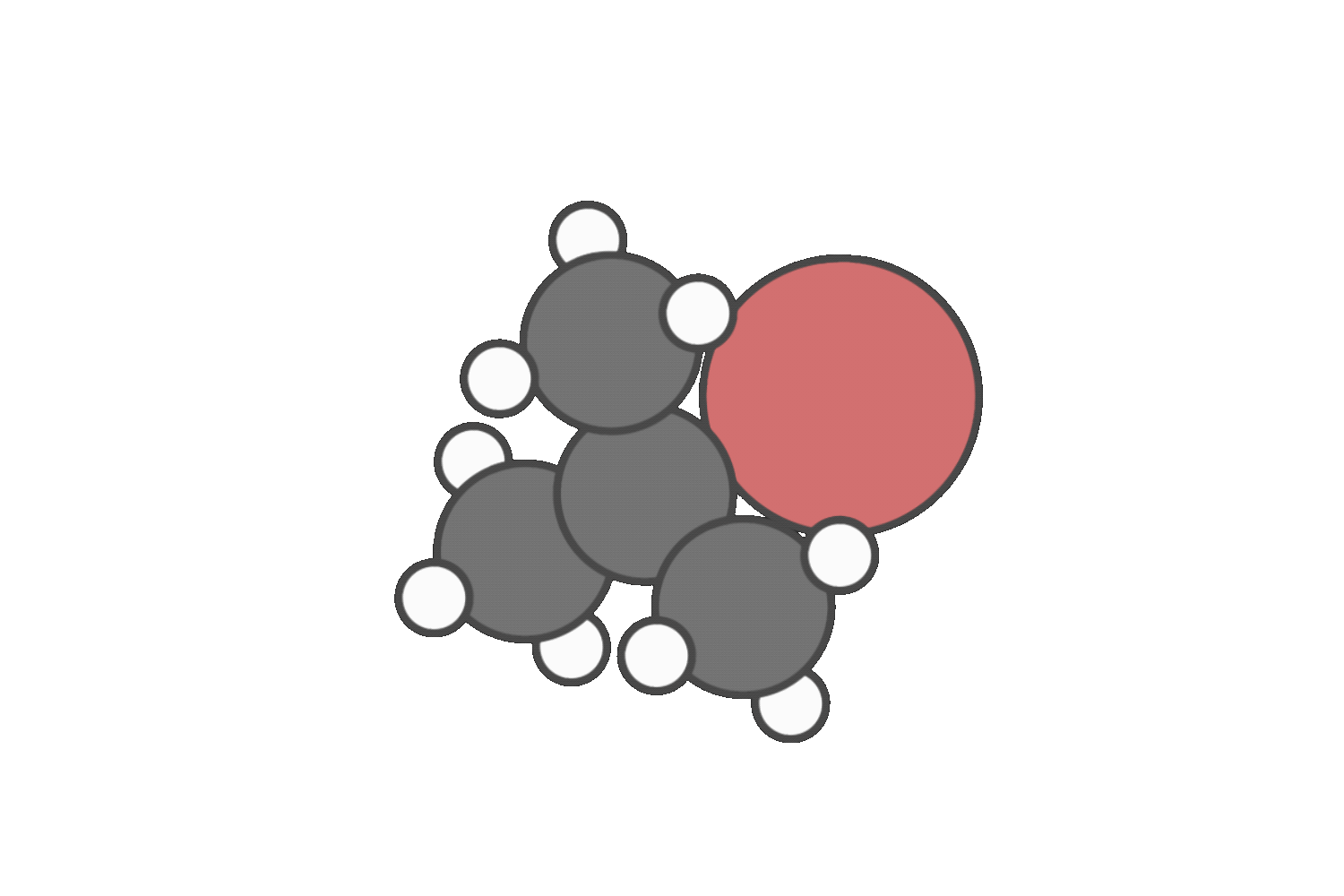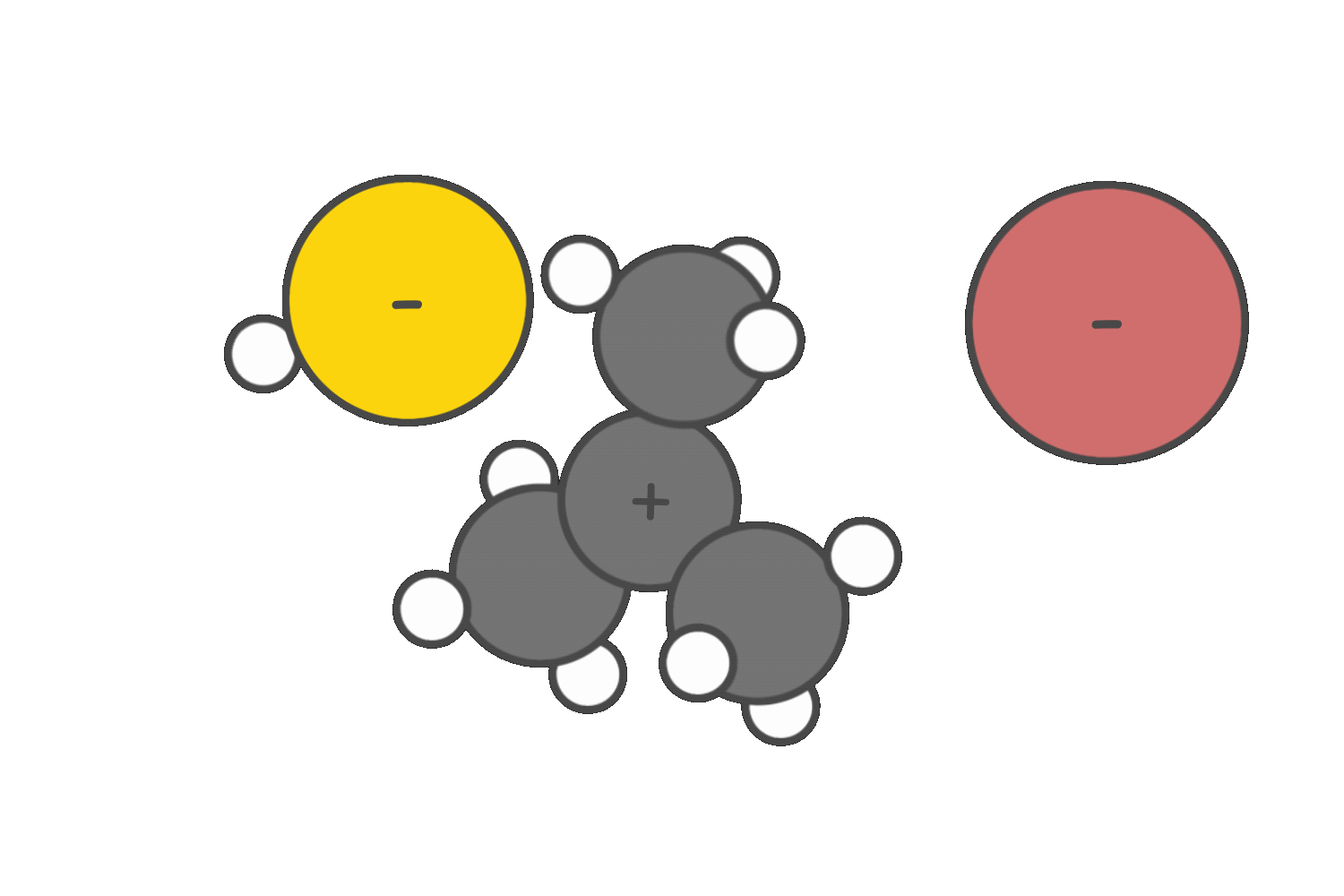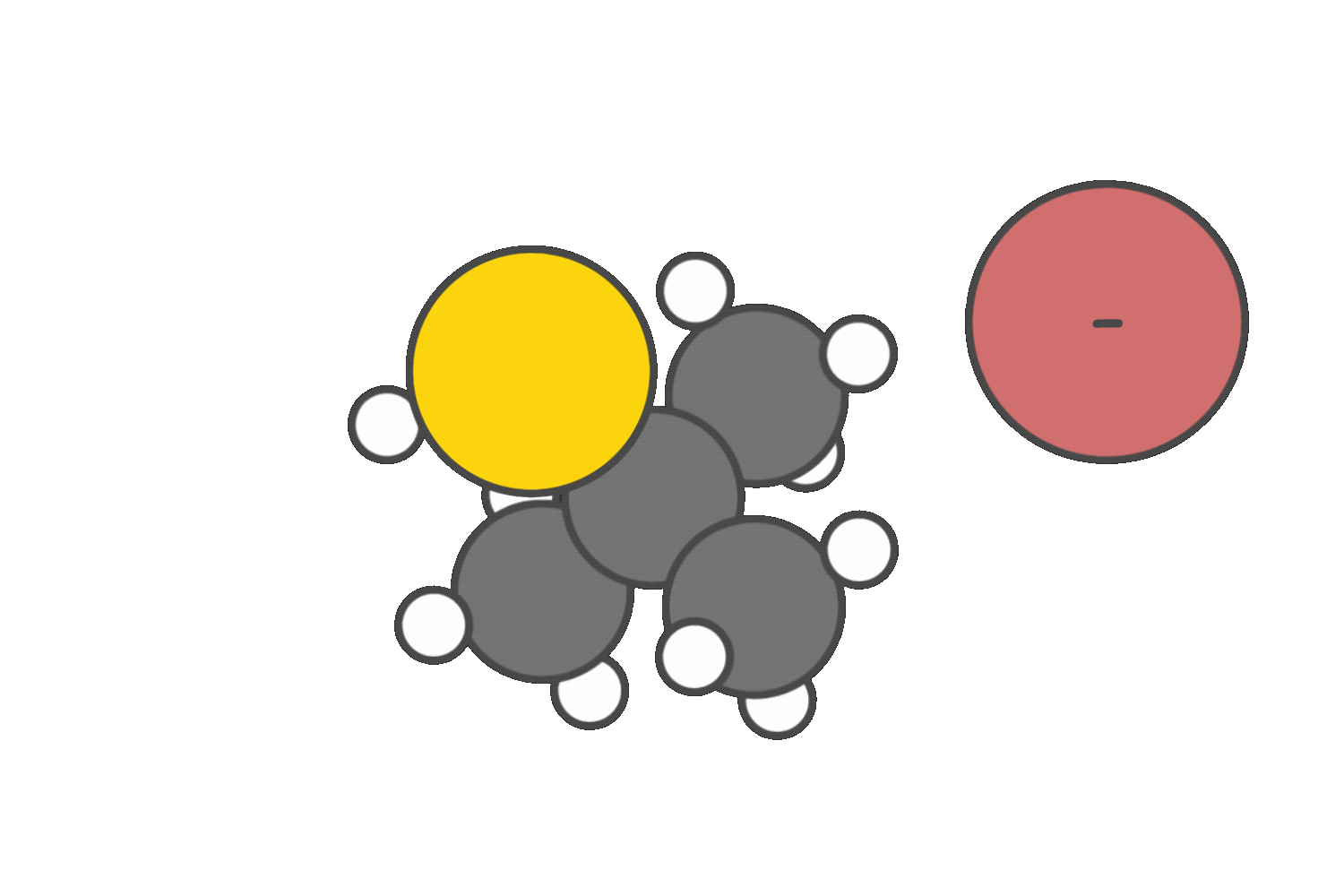
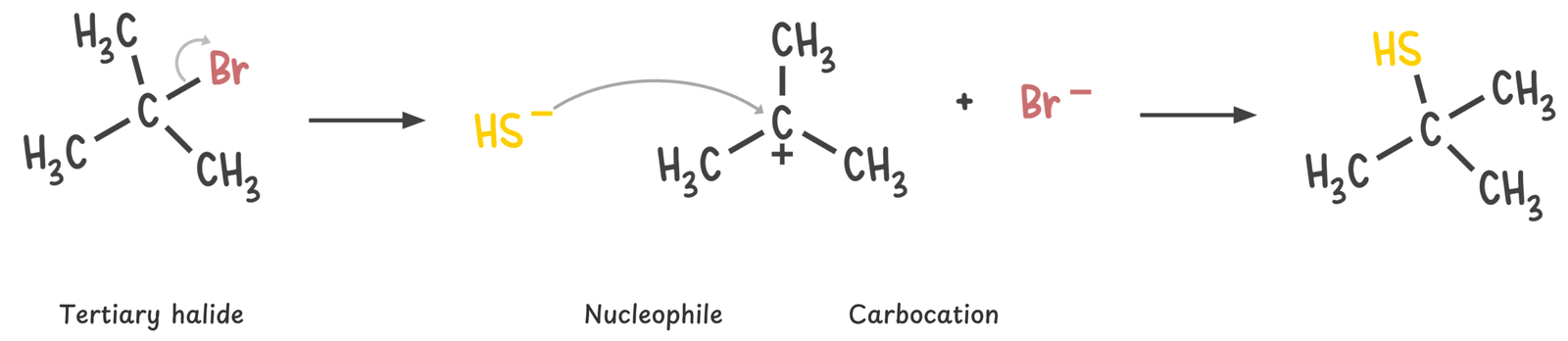
If a strong base such as CH3O– is present, elimination via the E2 mechanism becomes favorable, especially at higher temperatures.
Due to severe steric hindrance around the electrophilic center, SN2 is not feasible, and the strong base promotes β-hydrogen abstraction instead. The E2 reaction proceeds in a single concerted step, forming an alkene without generating a carbocation intermediate.
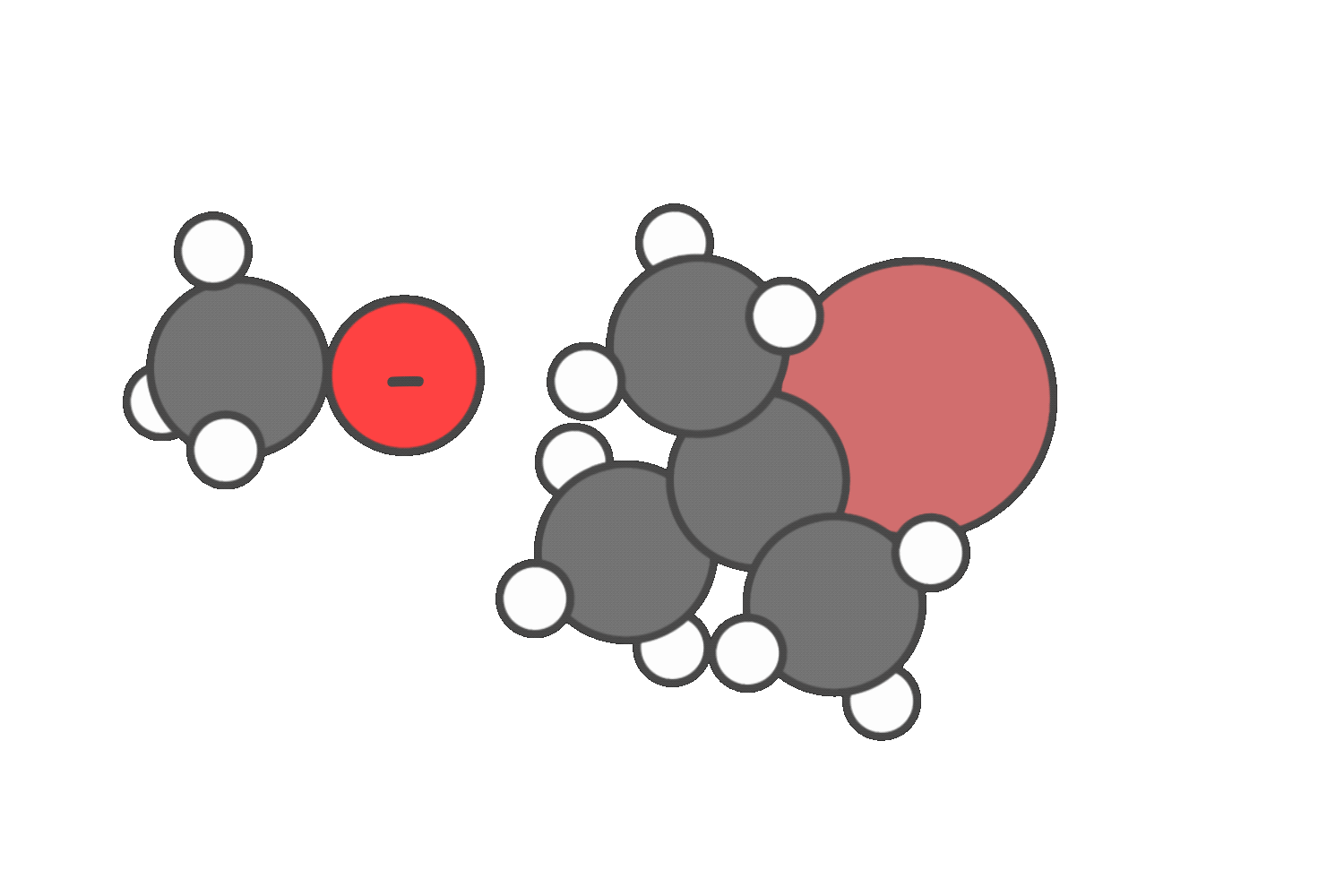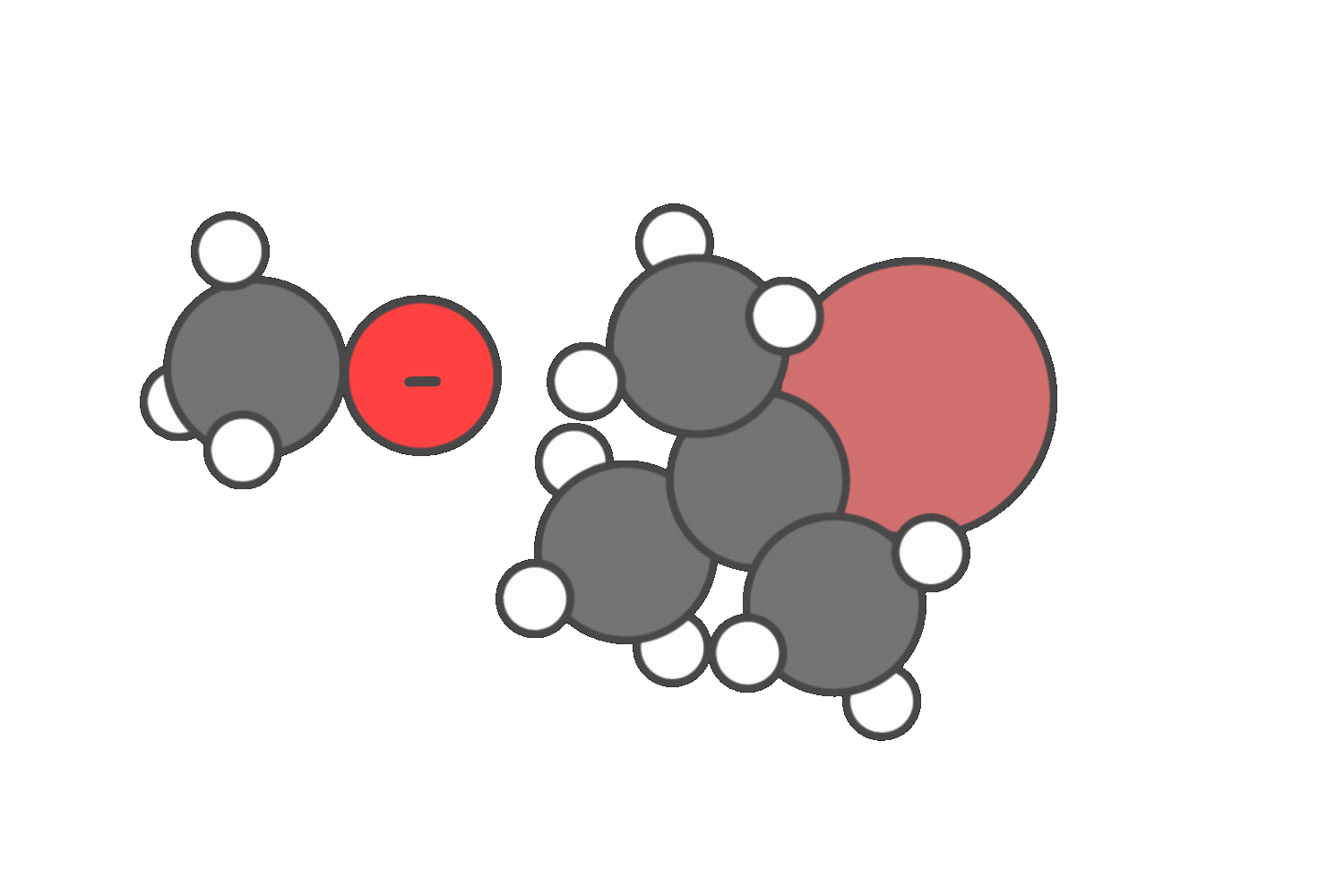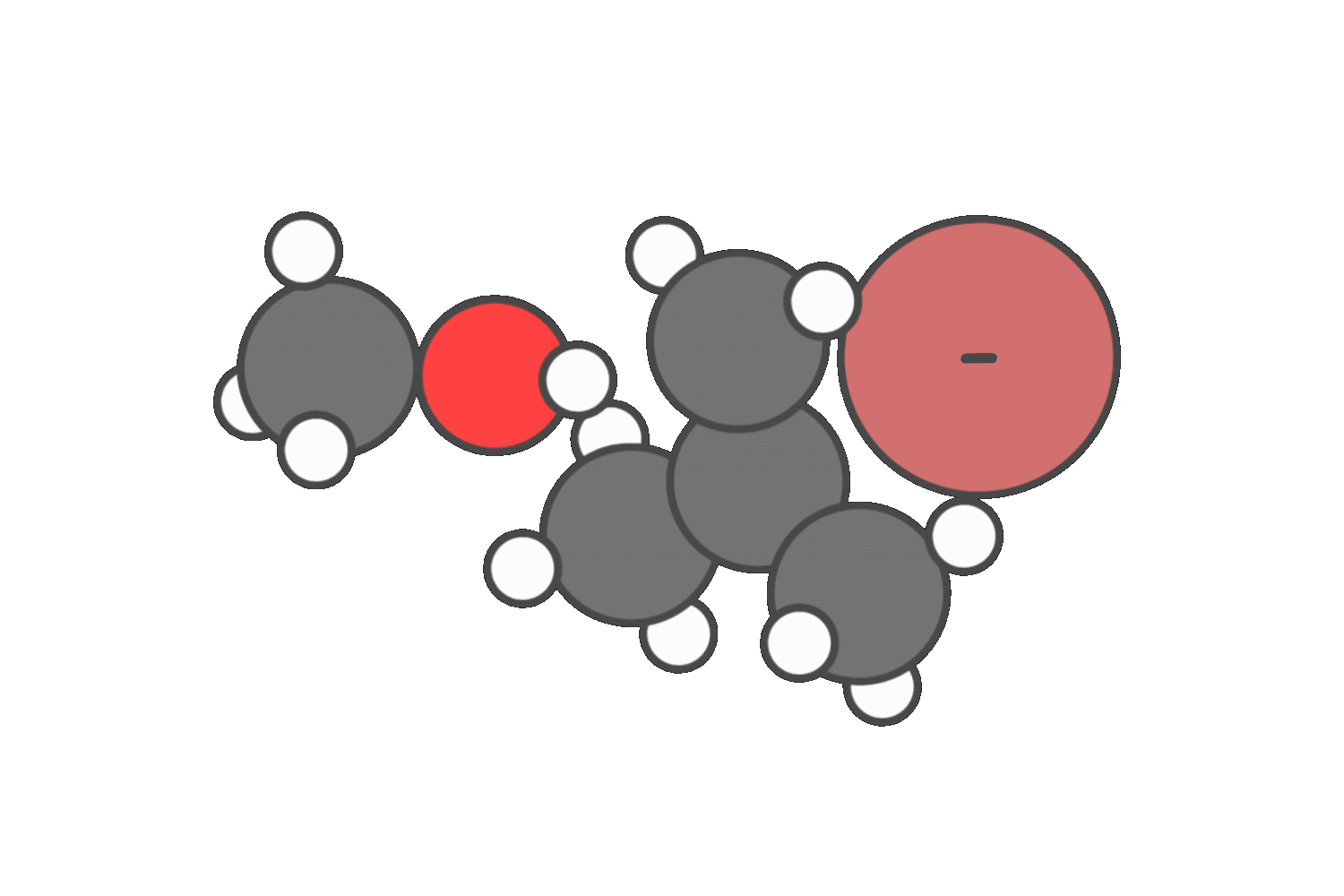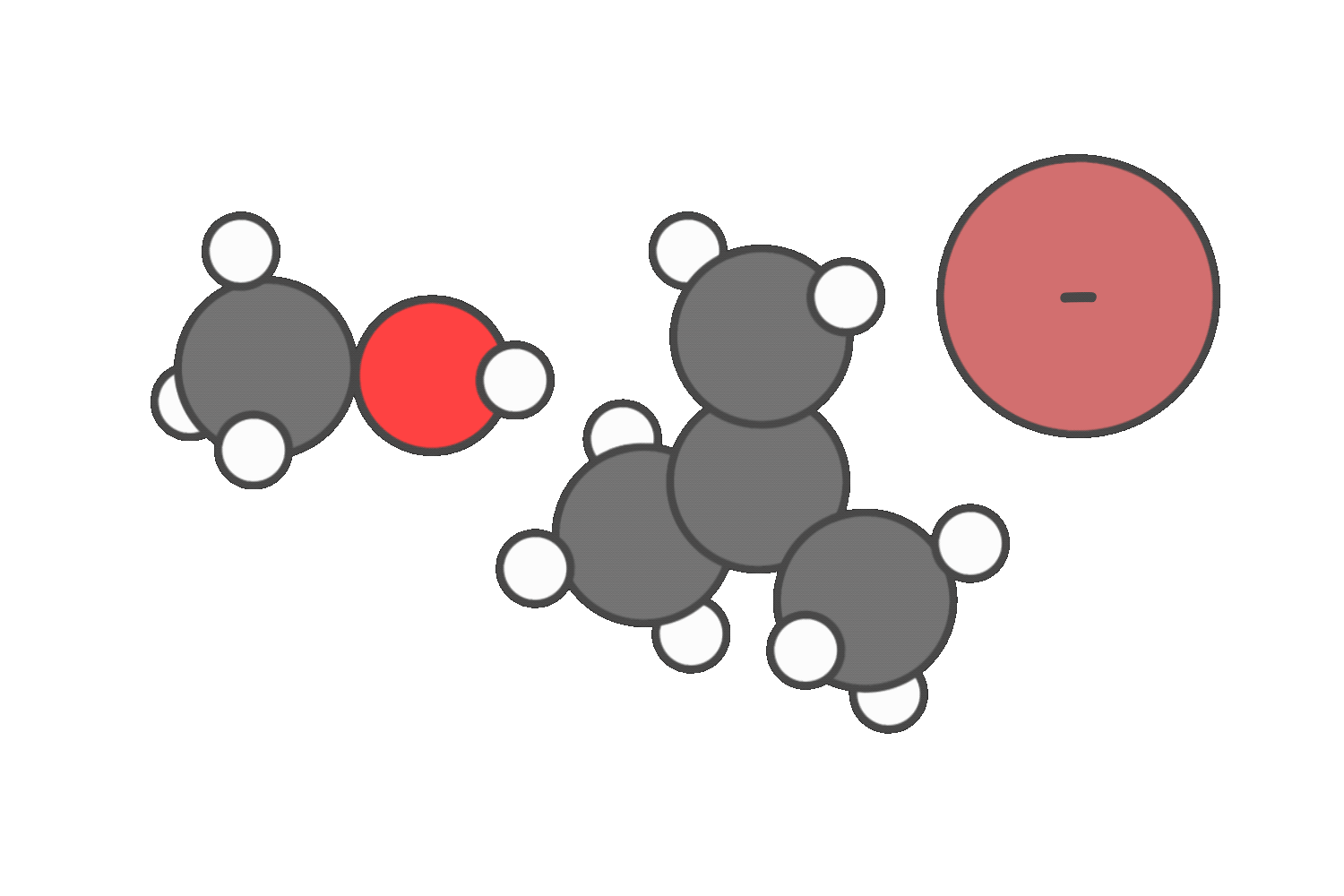

In neutral solvents like water or alcohols, both SN1 and E1 are possible. In these cases, you’ll often see a mixture of substitution and elimination products.
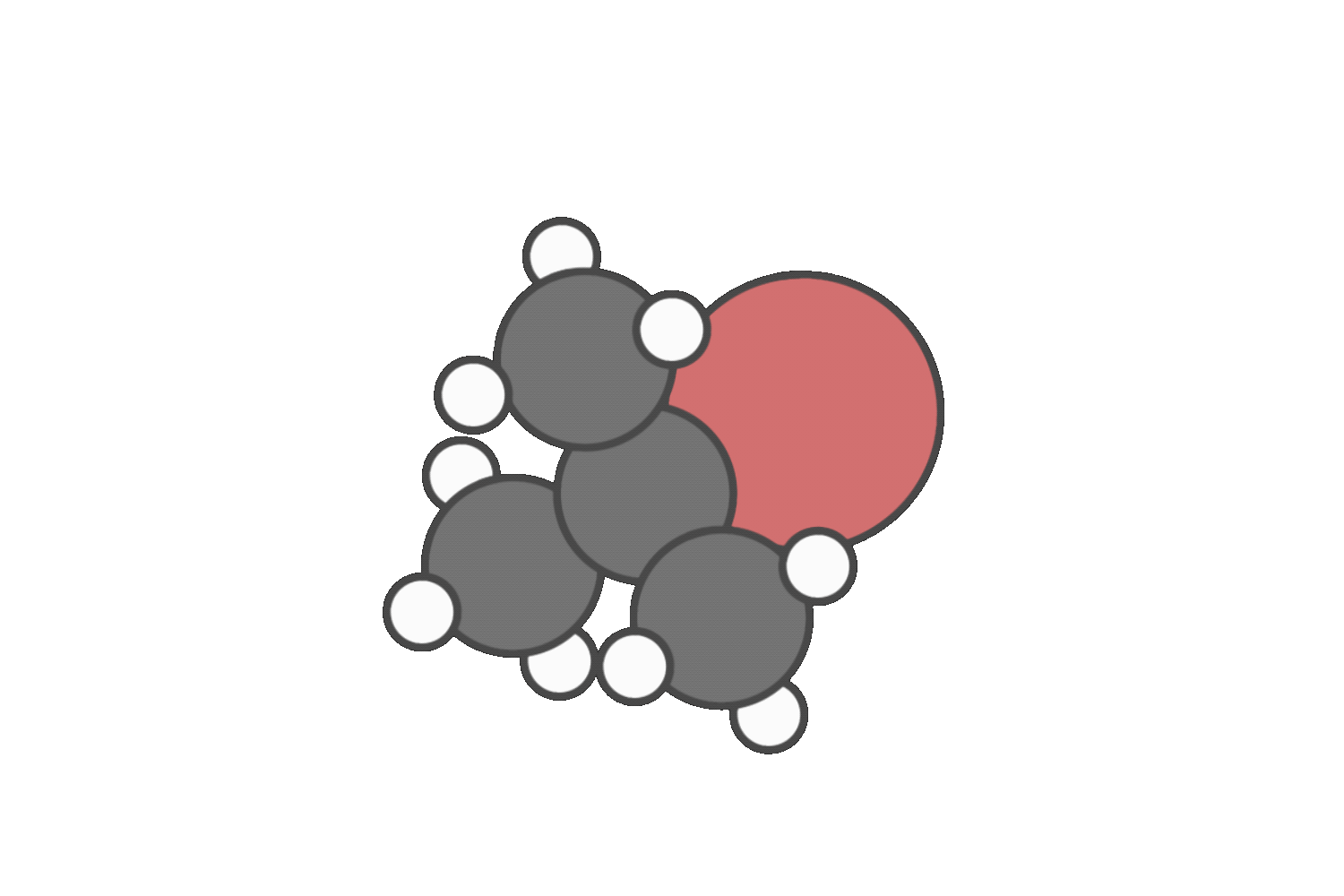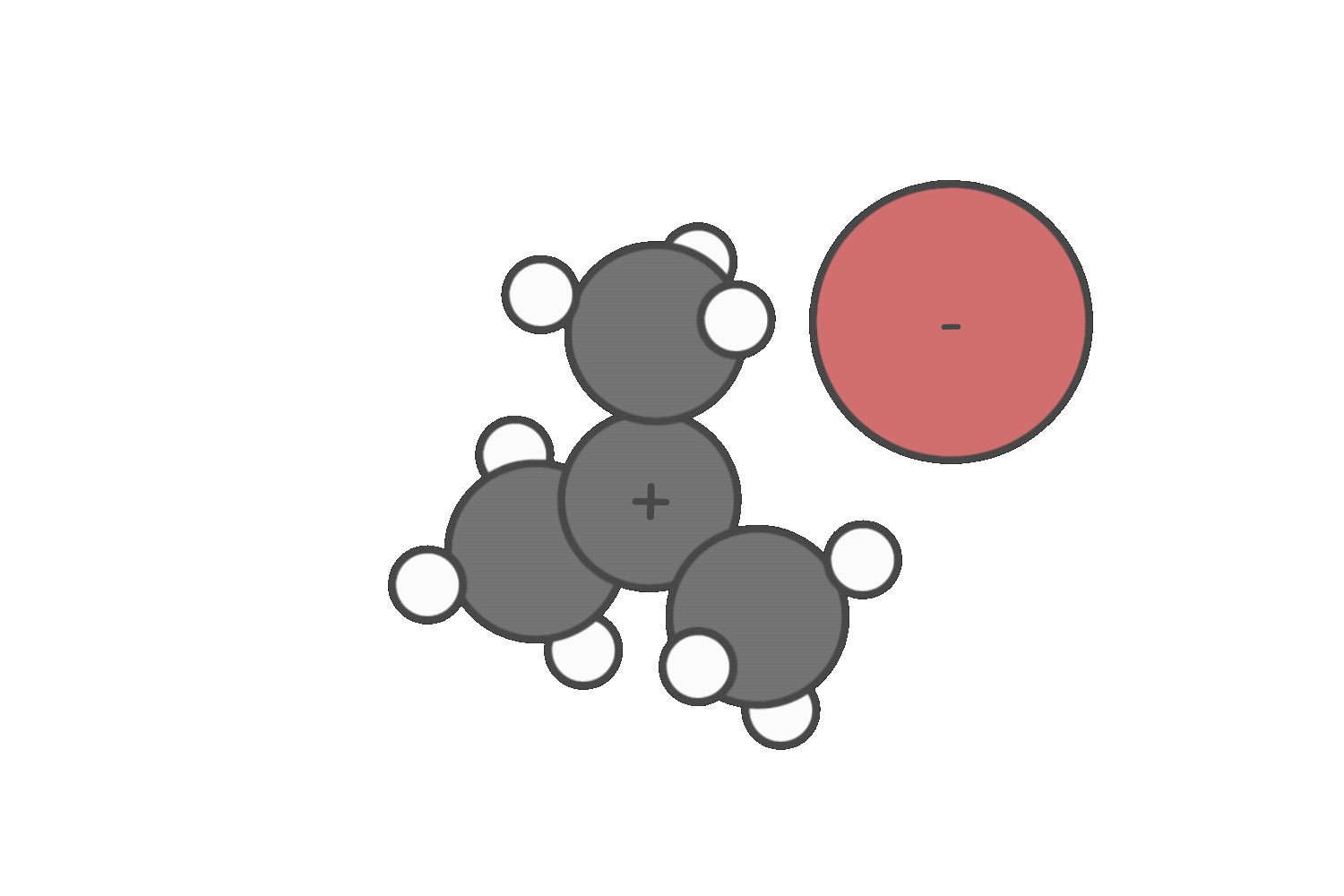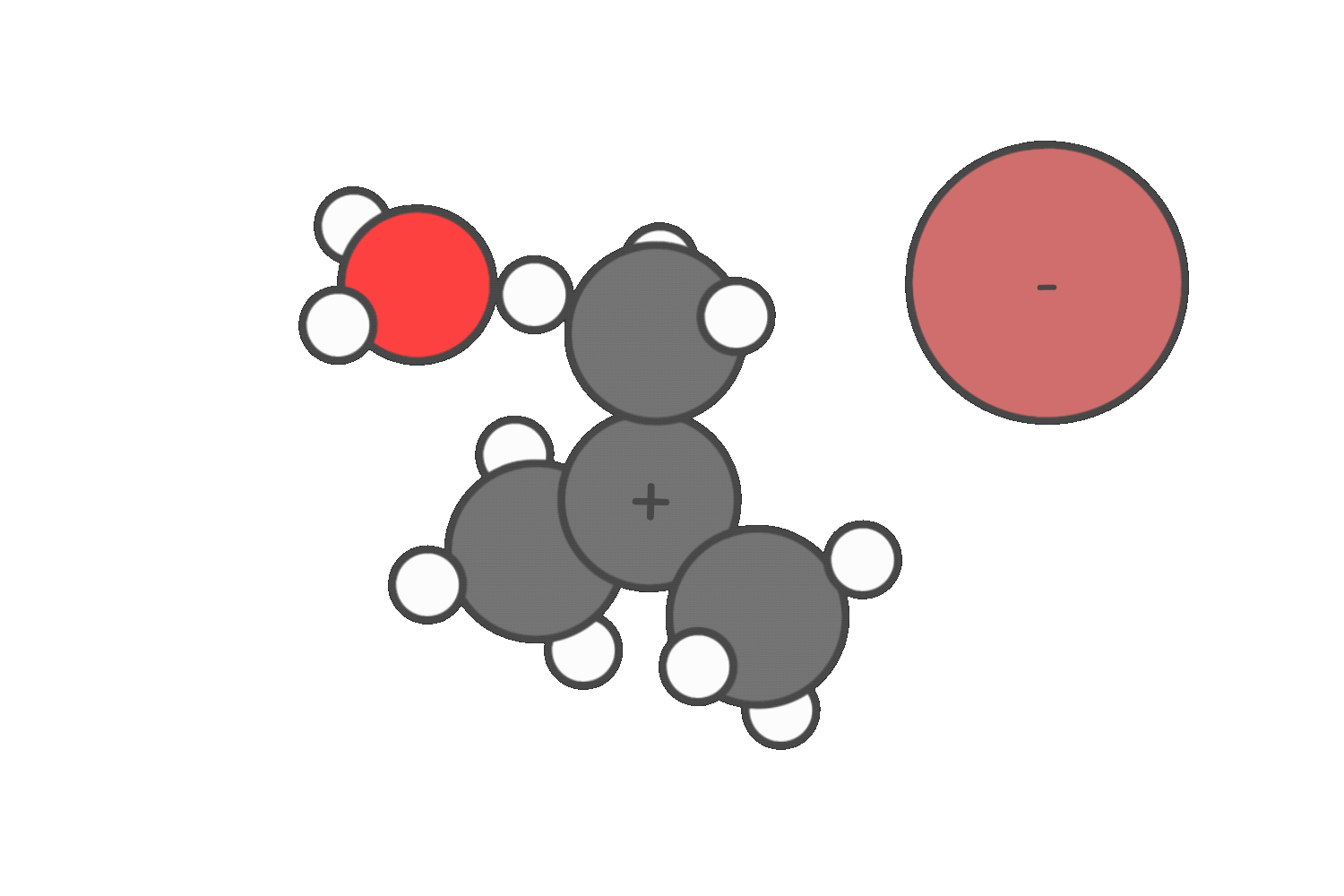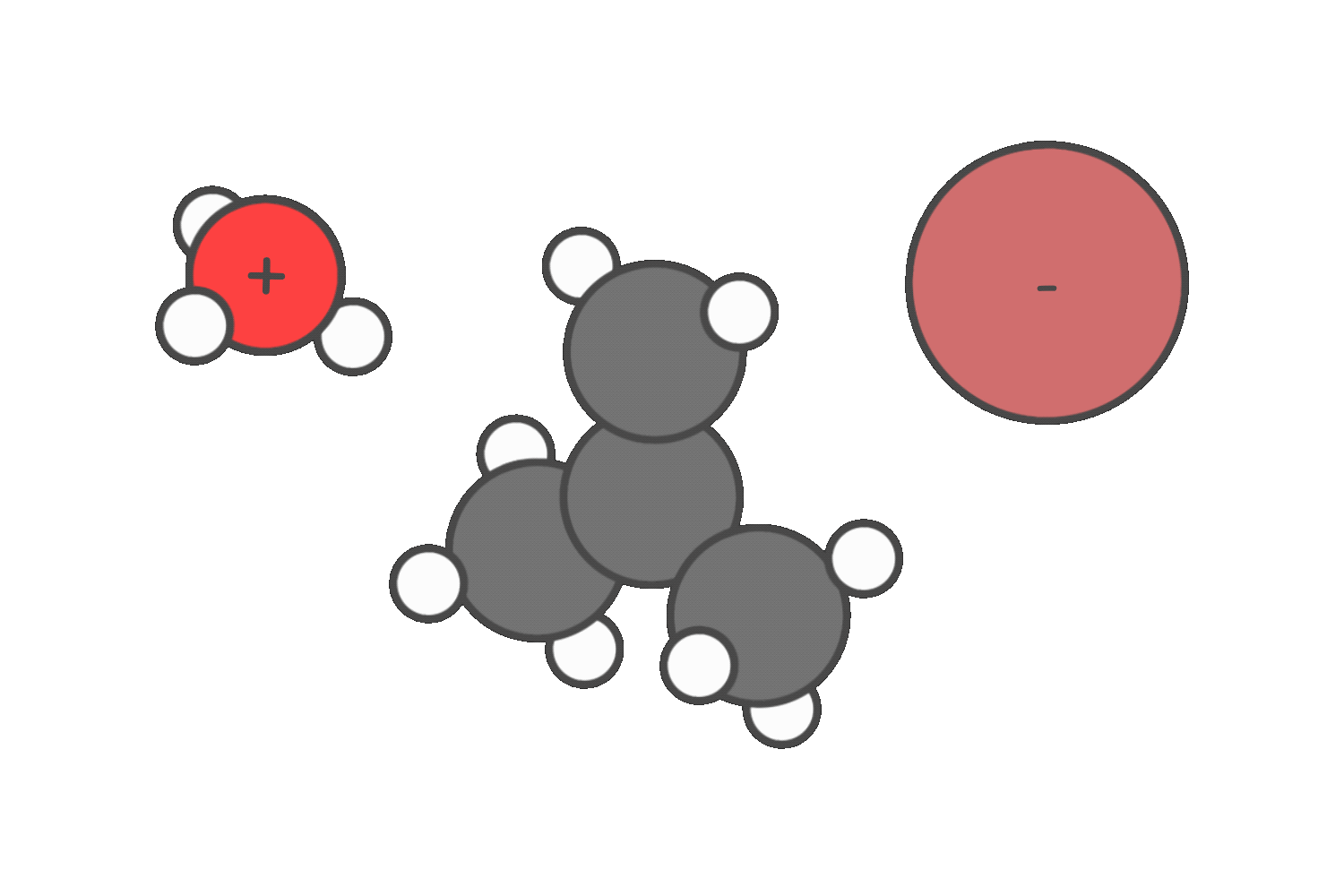
Step 2: Evaulate the nucleophile or base
In Step 1, we categorized some molecules as strong nucleophiles, others as strong bases, and a few as weak in both respects. Now we’ll take a closer look at what those labels actually mean, and how they influence the reaction outcome.
Start by asking: What kind of reagent am I working with? Is it a nucleophile, a base, or both? And is it strong or weak?
Nucleophiles are electron-rich species that attack the electrophilic carbon bonded to the leaving group. This leads to substitution reactions.

Bases, on the other hand, remove β-hydrogens from the substrate. This triggers elimination reactions.

The strength and identity of your reagent play a major role in determining the dominant reaction pathway.

Step 3: Consider temperature
Temperature tips the balance between substitution and elimination.
Ask: Is the reaction heated?
Heat promotes elimination. If you’re running a reaction at elevated temperatures, and there is a base present (even a weak one), E1 or E2 becomes more likely.
In contrast, substitution reactions often happen at room temperature or lower.
Step 4: Check the solvent
While the solvent usually doesn’t change the type of reaction that occurs, it can dramatically affect the reaction rate and efficiency.
Why this matters:
SN1 and E1 reactions proceed through a carbocation intermediate. Polar protic solvents, such as water or alcohols, help stabilize this positively charged species by surrounding it and donating electron density through their lone pairs.
This stabilization lowers the energy barrier for carbocation formation, making these reactions more favorable.
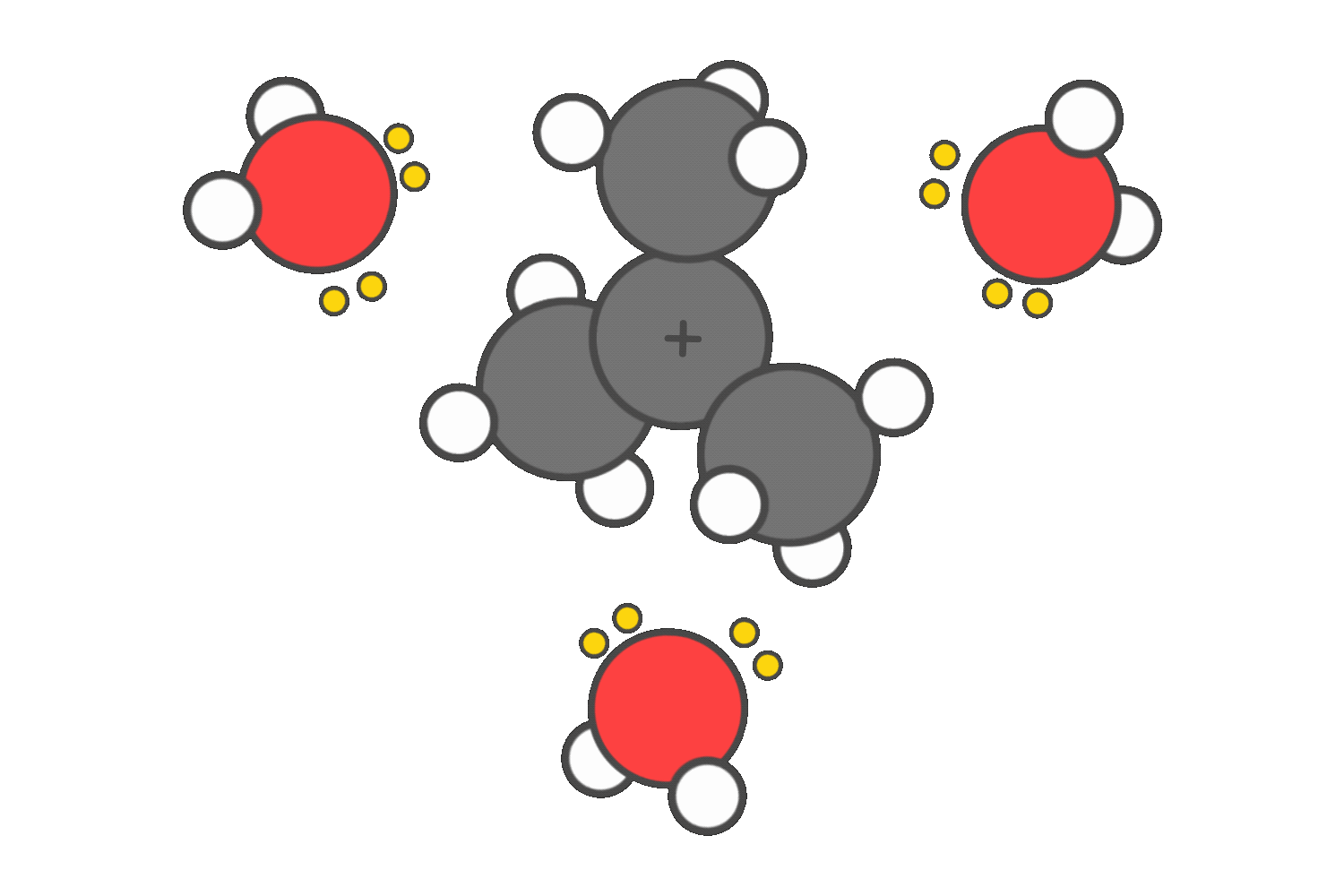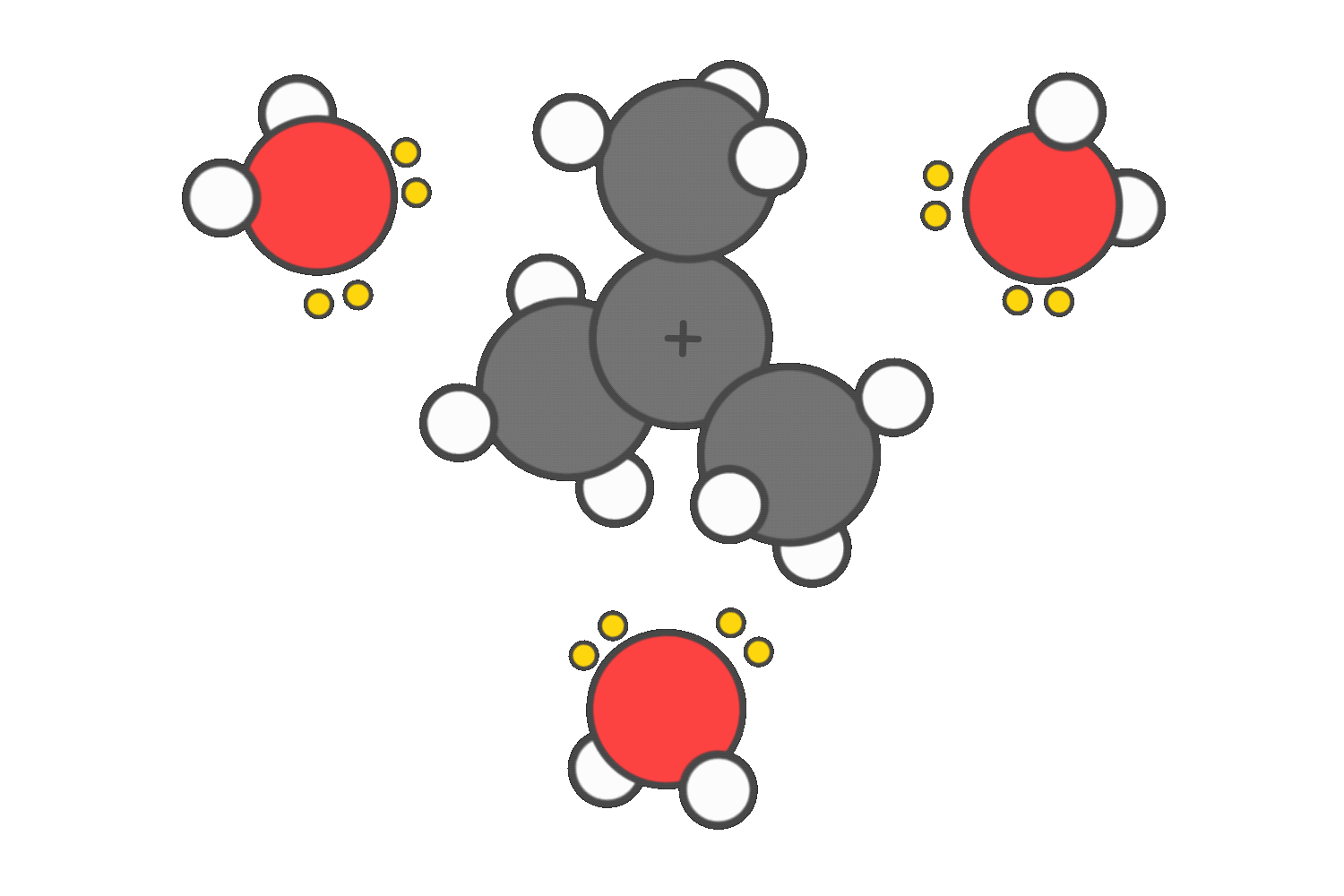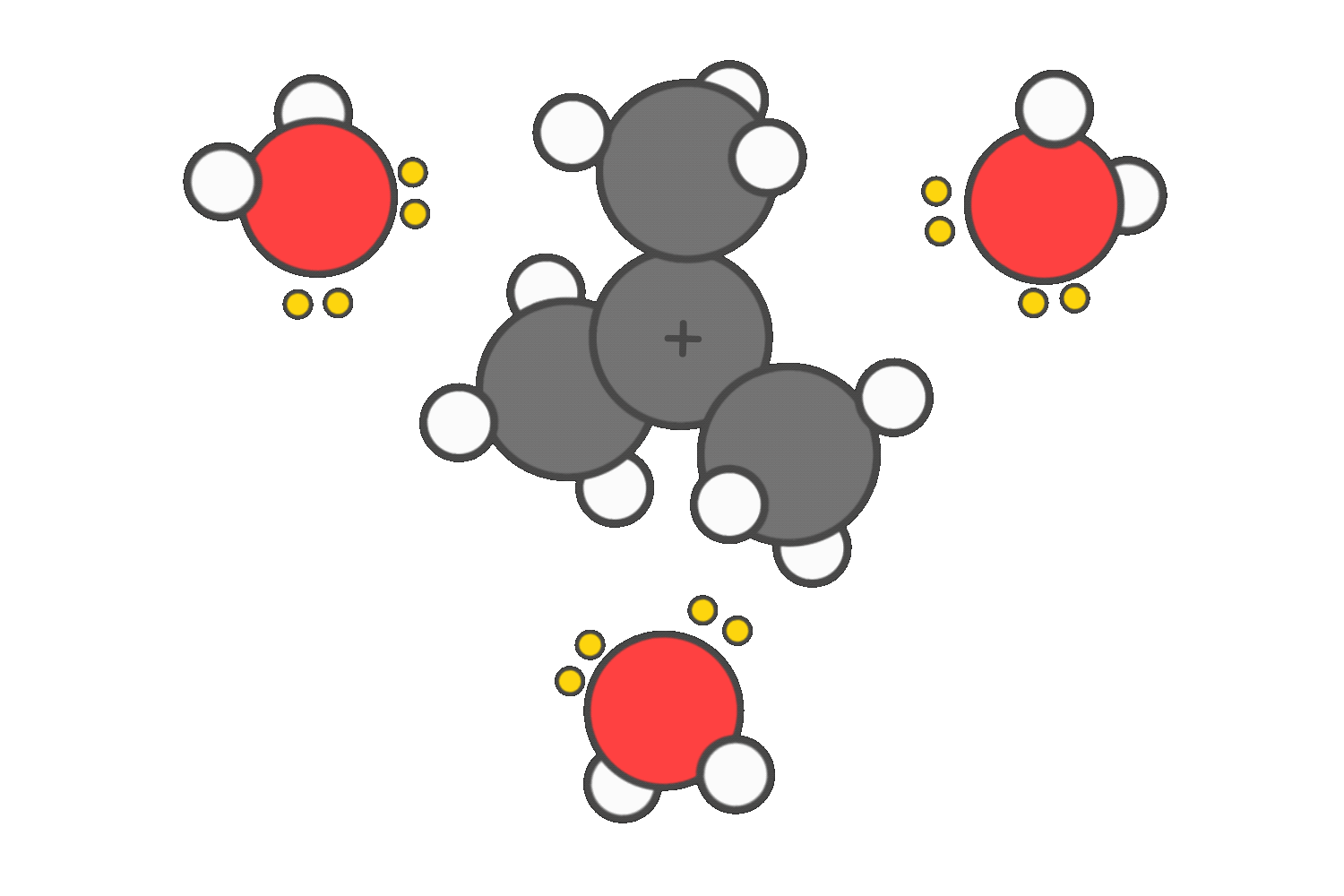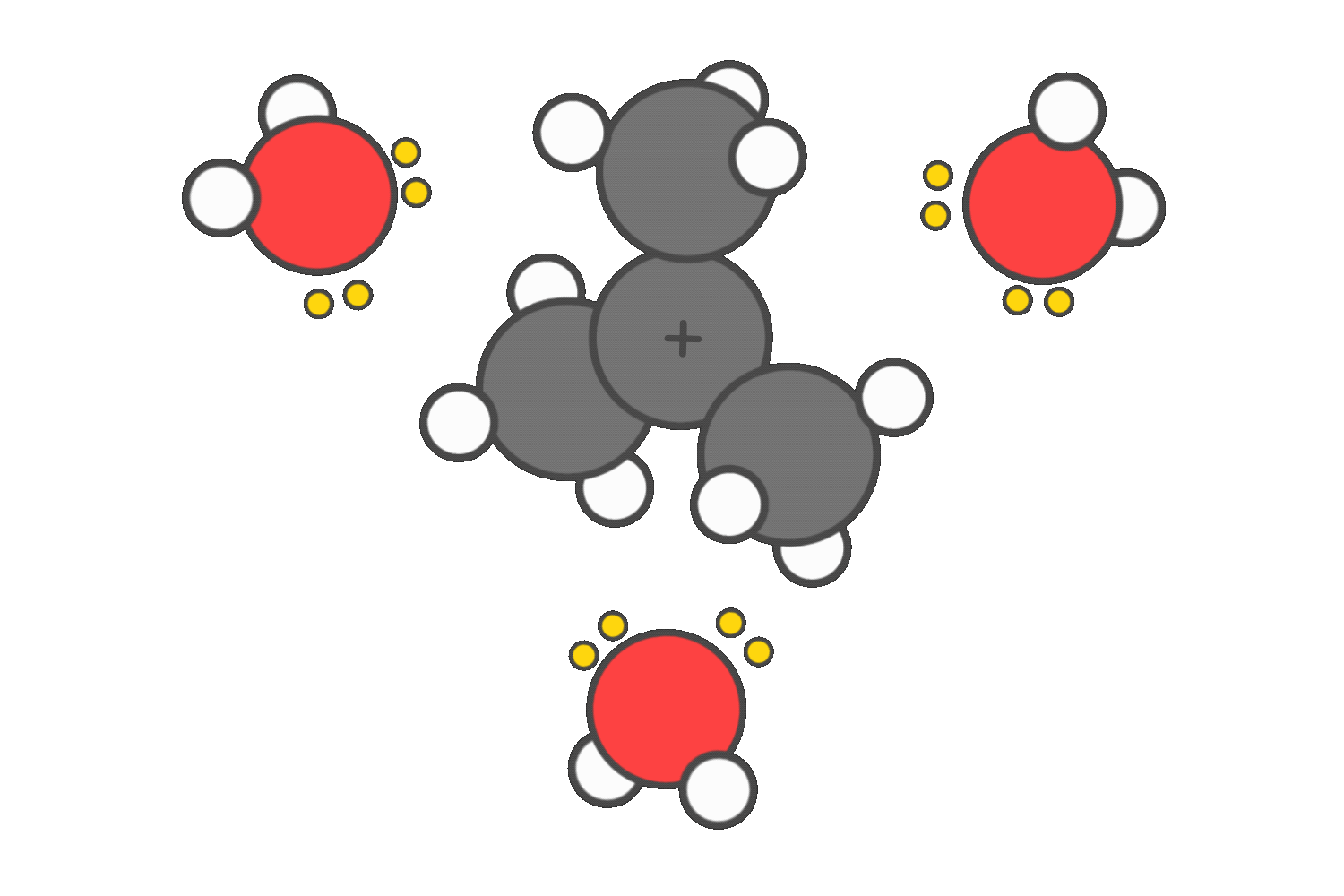
In contrast, SN2 reactions are slowed by polar protic solvents. These solvents form strong hydrogen bonds with the nucleophile, effectively trapping it and reducing its ability to attack the electrophilic carbon.
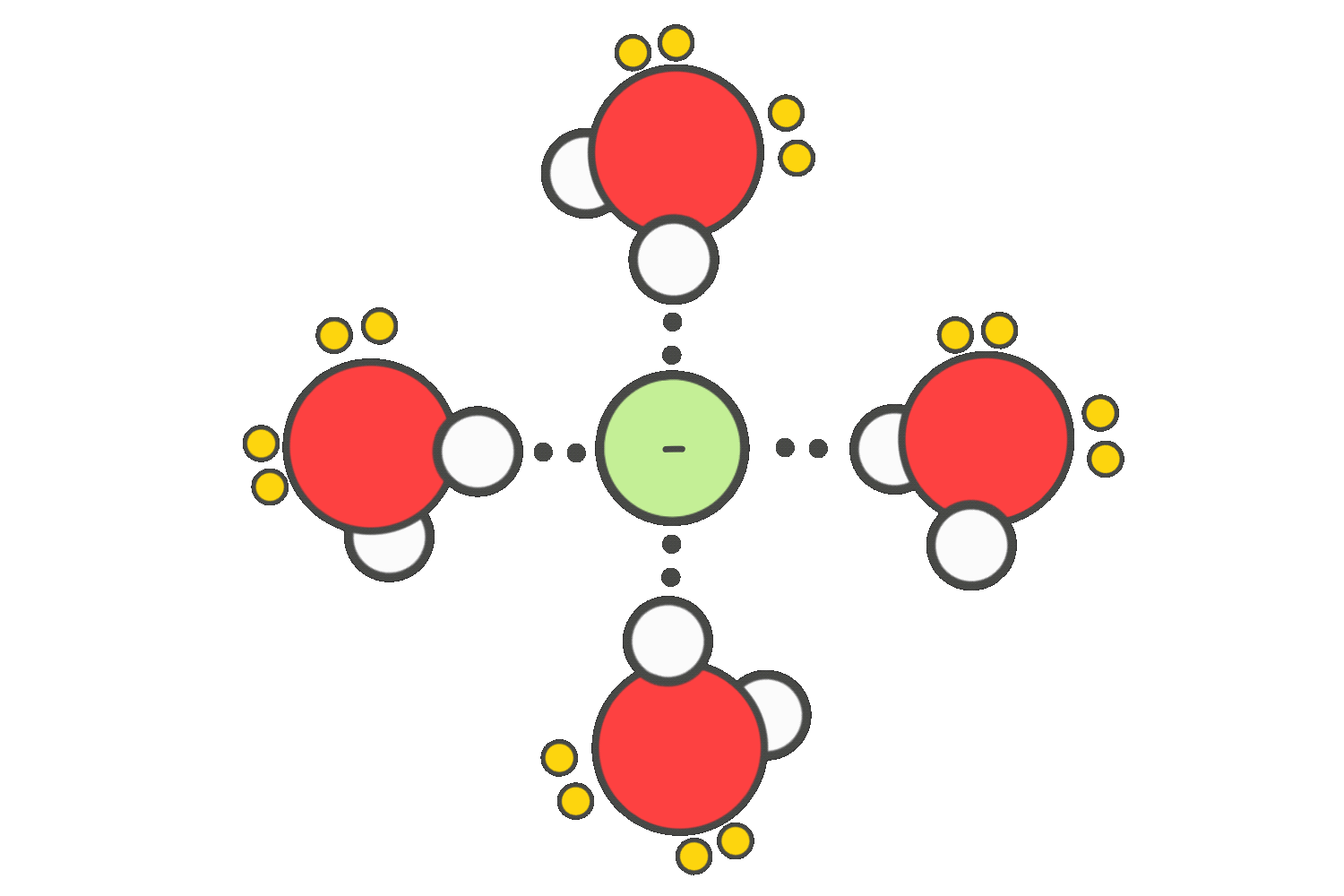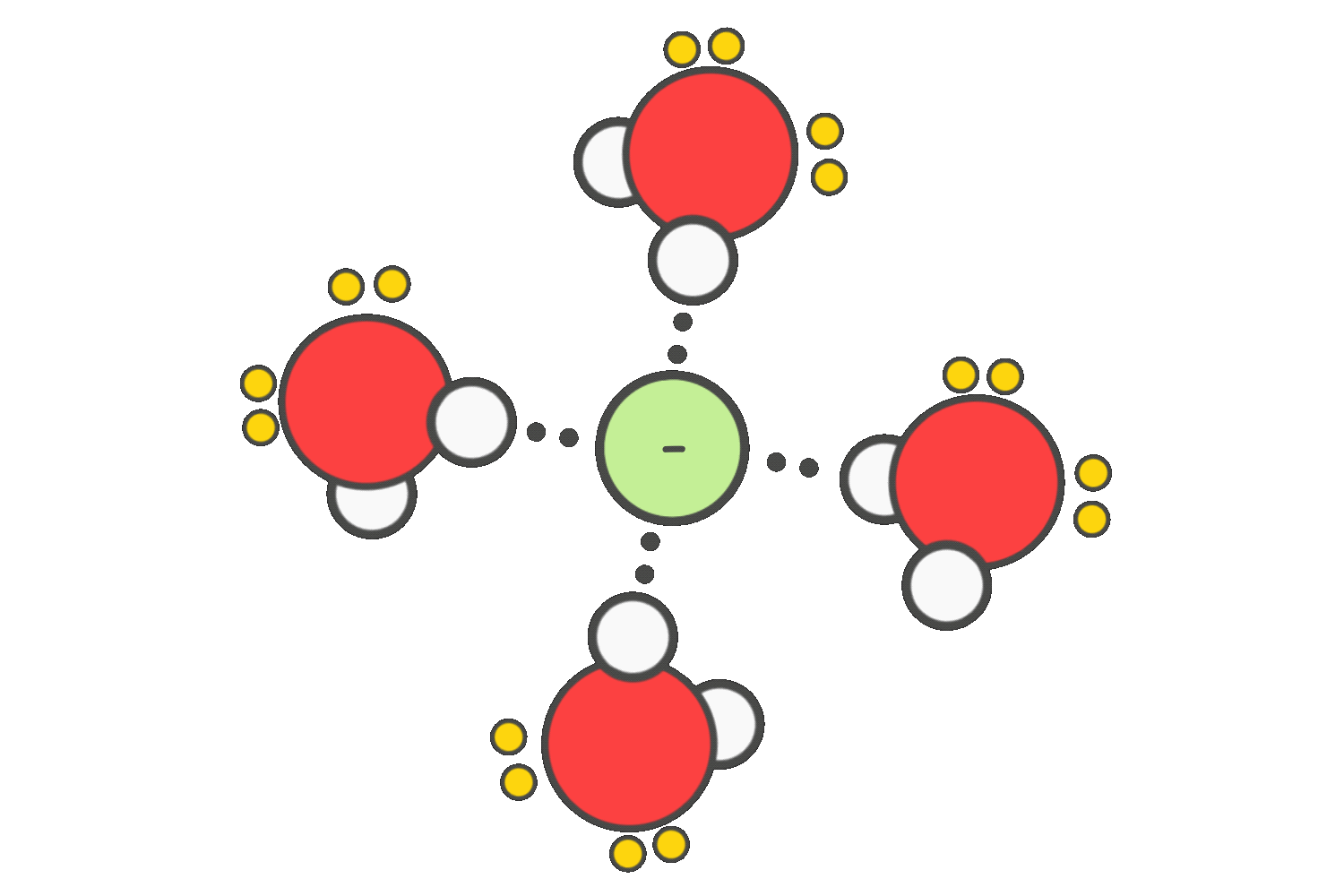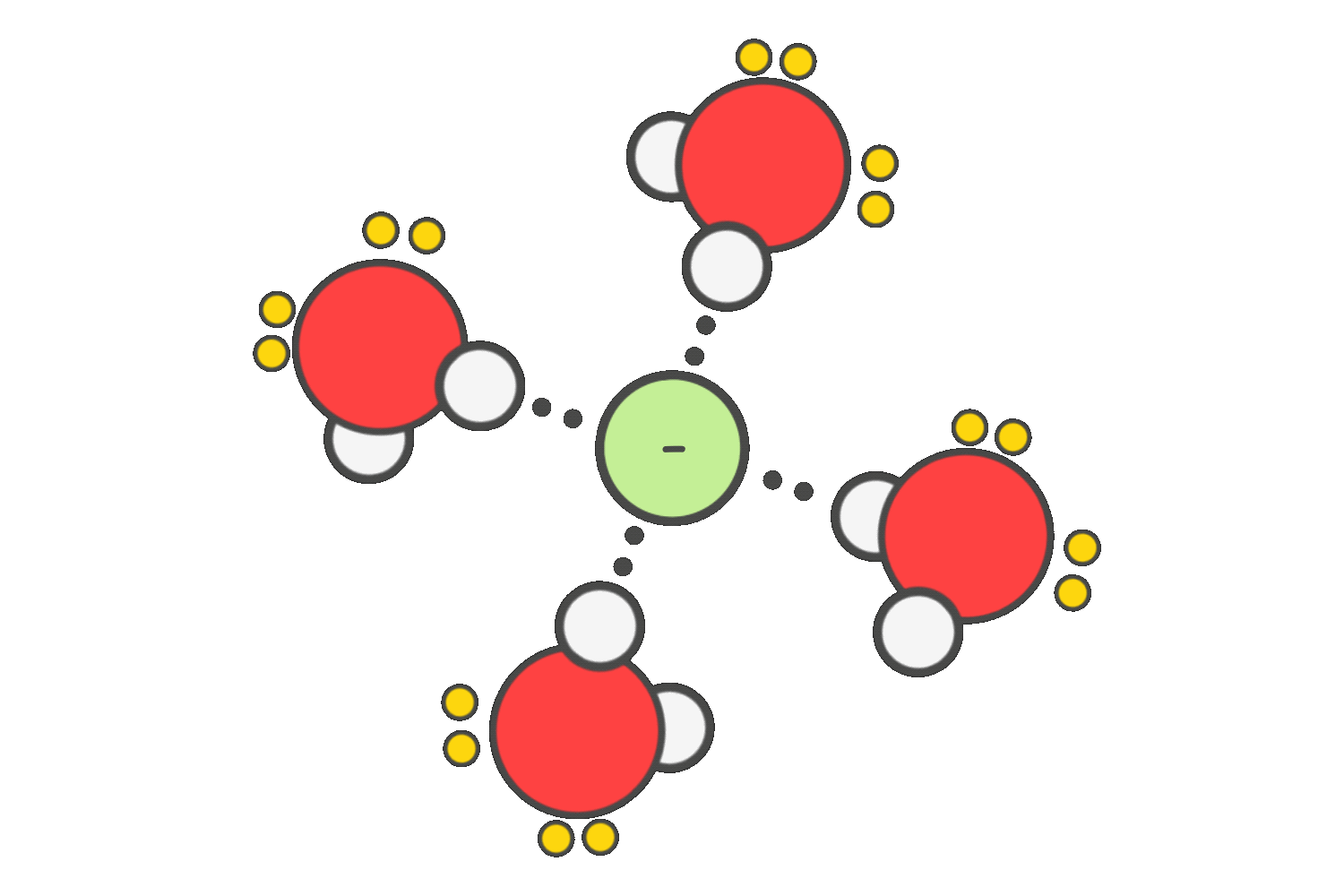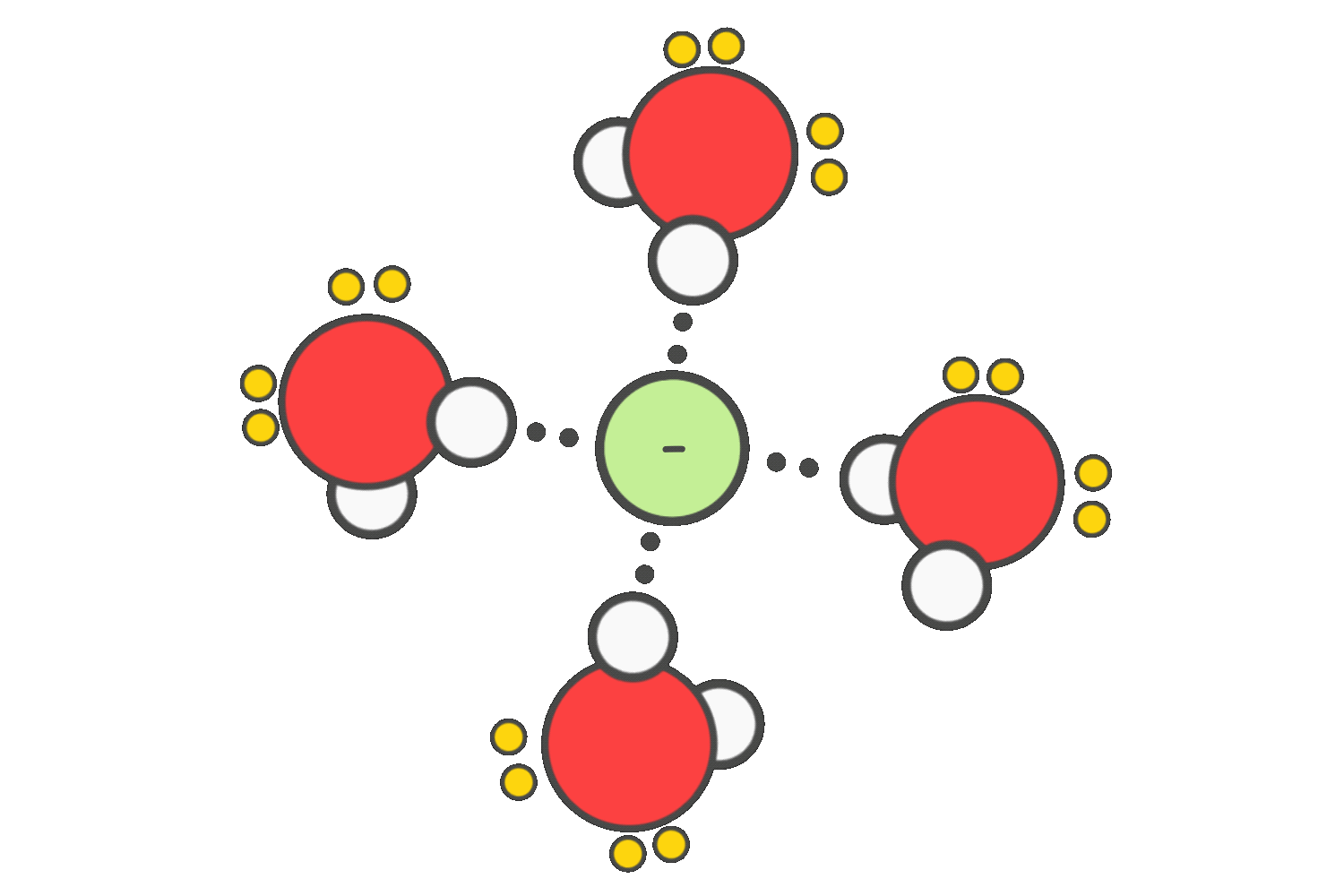
To promote SN2 reactions, polar aprotic solvents such as acetone, DMSO, or DMF are preferred. These solvents do not hydrogen-bond with the nucleophile, allowing it to remain “naked” and highly reactive, which increases the reaction rate significantly.
Putting it all together
You don’t need to memorize a dozen mechanisms or flowcharts. You just need to ask the right questions.
- What kind of substrate do I have?
- What kind of nucleophile or base is present?
- Is the reaction heated?
- What solvent is used?
Once you answer these, you’ll usually be able to predict the correct pathway.
If a reaction looks like it could go multiple ways, remember that you may get a mixture of products. This is common with secondary substrates and mixed-function reagents (like hydroxide, which is both a base and a nucleophile).
So take it step by step, focus on principles rather than memorization, and keep practicing with real examples. Eventually, this process becomes second nature.
