Step 1: Classify the carbocation
First things first—identify the carbocation. Find the carbon atom with the positive charge. That’s the carbocation center.
Once you’ve located it, ask yourself:
How many carbon (alkyl) groups are directly bonded to this positively charged carbon?
Carbocations are classified as methyl, primary (1°), secondary (2°), or tertiary (3°) based on the number of alkyl groups connected to the carbocation center.
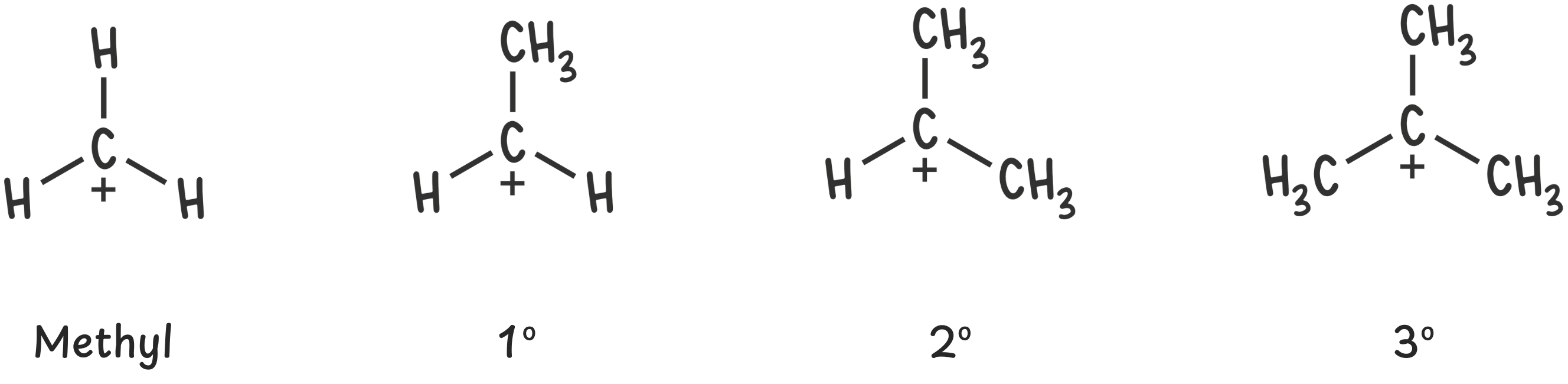
But there’s more!
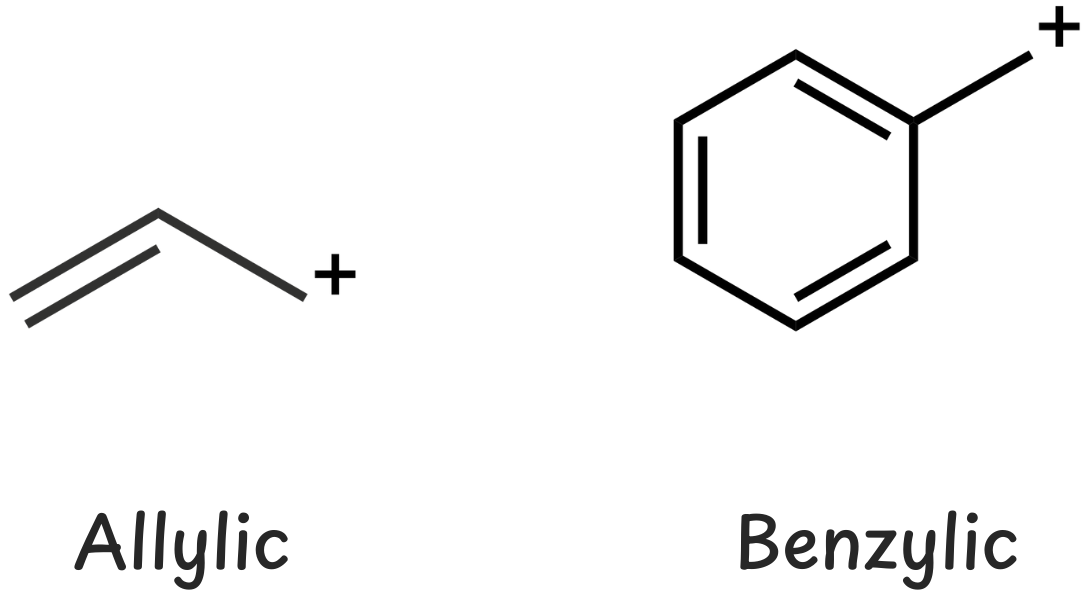
If the carbocation is next to:
- A double bond, it’s allylic.
- A benzene ring, it’s benzylic.
Step 2: Consider factors that stabilize carbocations
Now that you’ve classified the carbocations, it’s time to think about what makes some carbocations more stable than others. There are two big stabilizing factors: substitution and resonance. Let’s break them down.
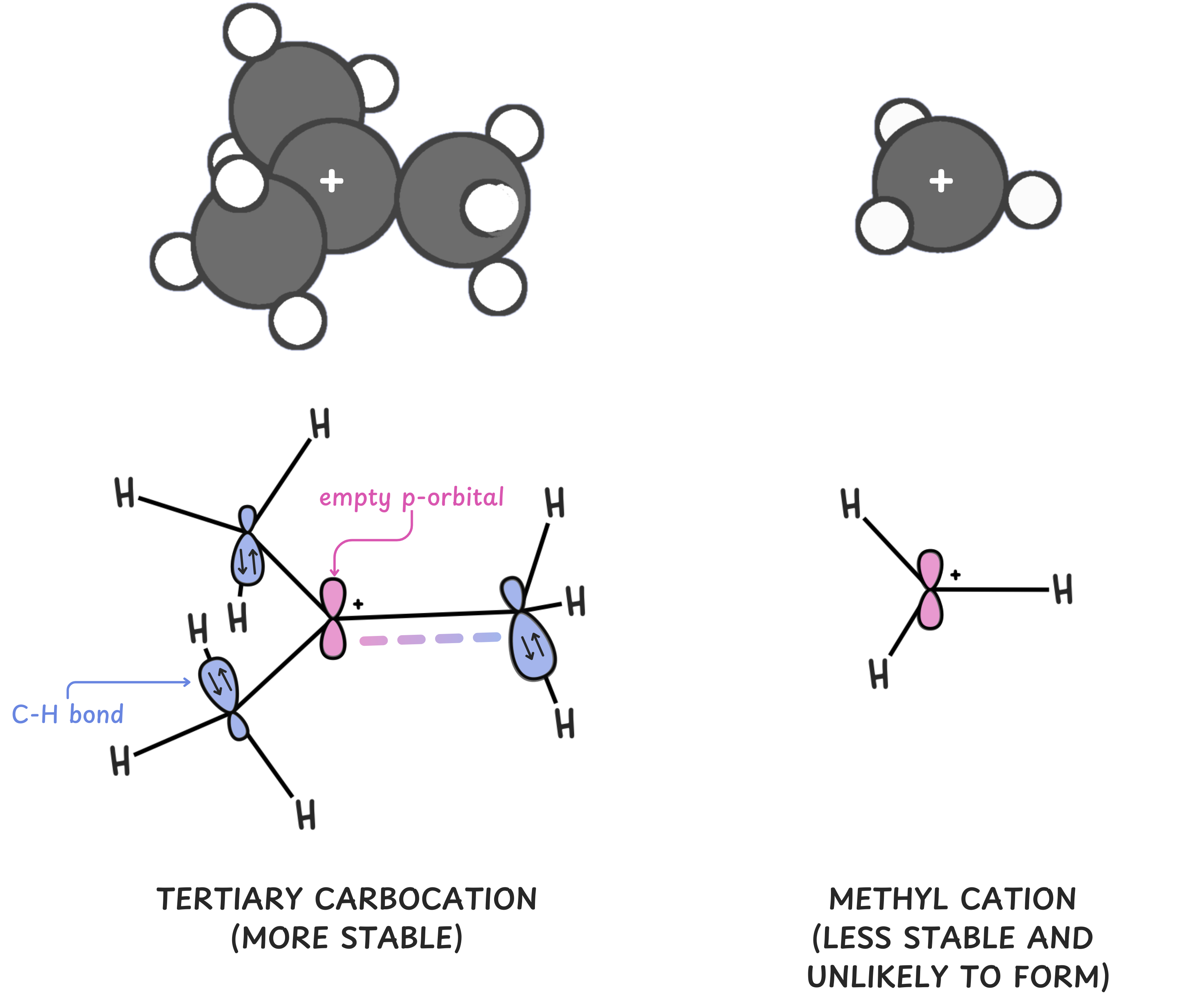
1️⃣ Alkyl substituents stabilize carbocations
Key rule: The more carbon substituents directly attached to the carbocation, the more stable it is.
Why? Two big reasons:
Hyperconjugation: Neighboring C–H bonds can overlap slightly with the empty p-orbital of the carbocation, donating electron density to help stabilize the positive charge.
Inductive Effect: Alkyl groups push electron density toward the carbocation, reducing the charge density.
As a result, we get the following trend for carbocation stability.
Methyl << Primary (1°) < Secondary (2°) < Tertiary (3°)
2️⃣ Adjacent C-C pi bonds stabilize carbocations
Key rule: Allylic and benzylic carbocations are more stable than regular carbocations with the same number of alkyl substituents.
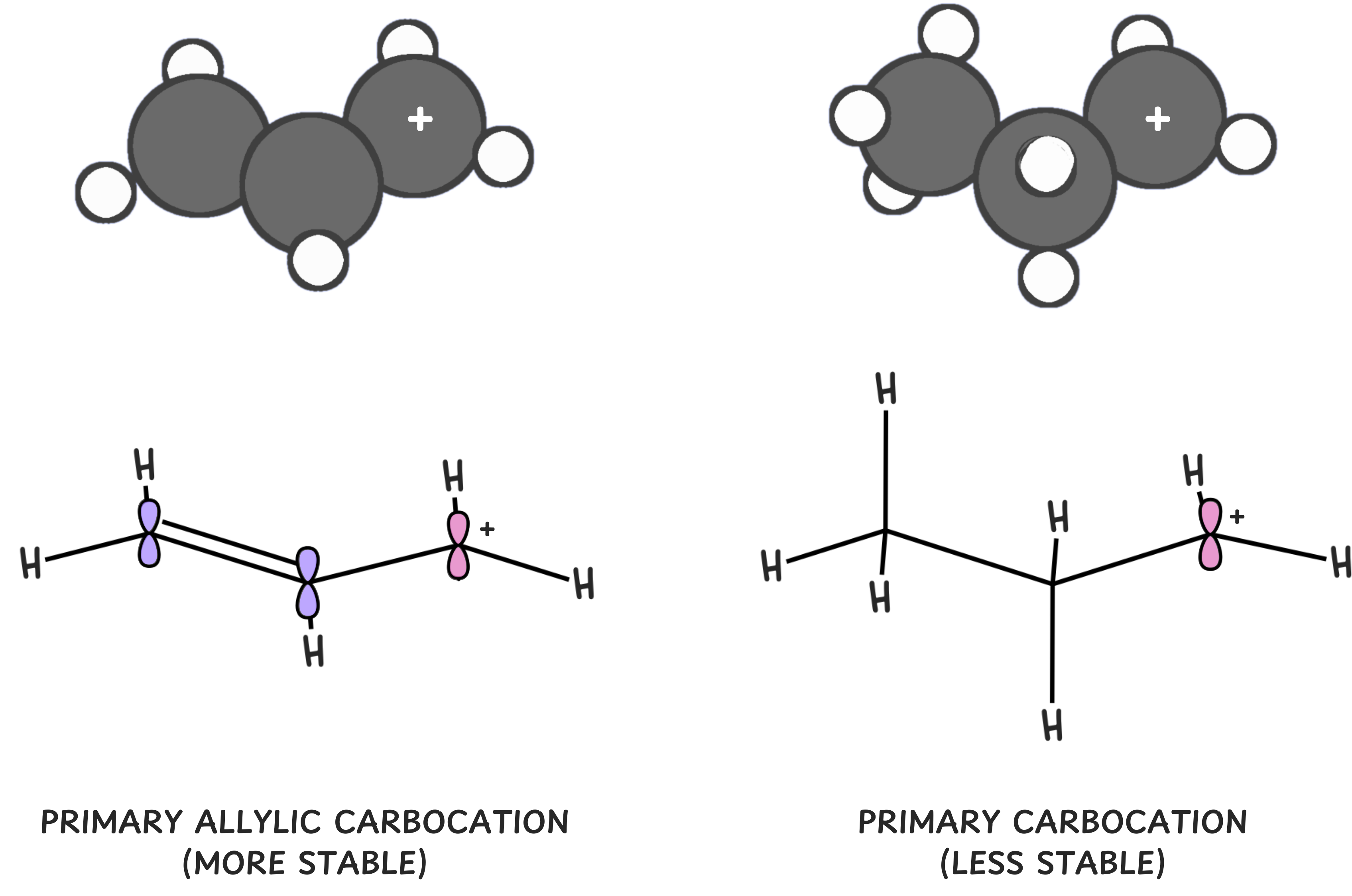
Why? The positive charge in allylic and benzylic carbocations is delocalized (spread out) across multiple atoms, reducing the charge density and stabilizing the carbocation.
3️⃣ Adjacent lone pairs stabilize carbocations
If an atom next to the carbocation has lone pairs, it can donate electrons via resonance.
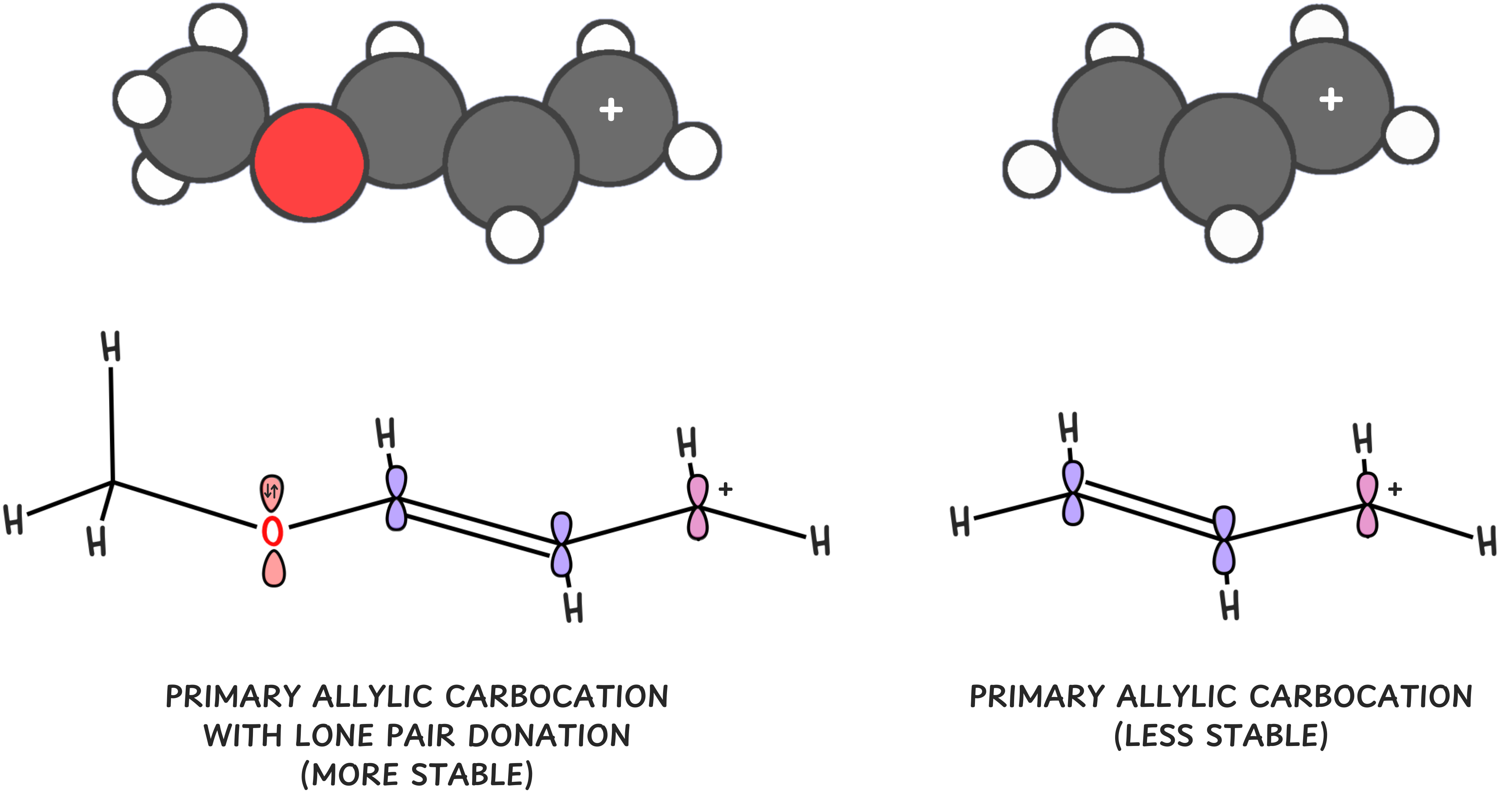
Those lone pairs can resonate into the empty p-orbital of the carbocation, giving it a full octet and significantly increasing stability.
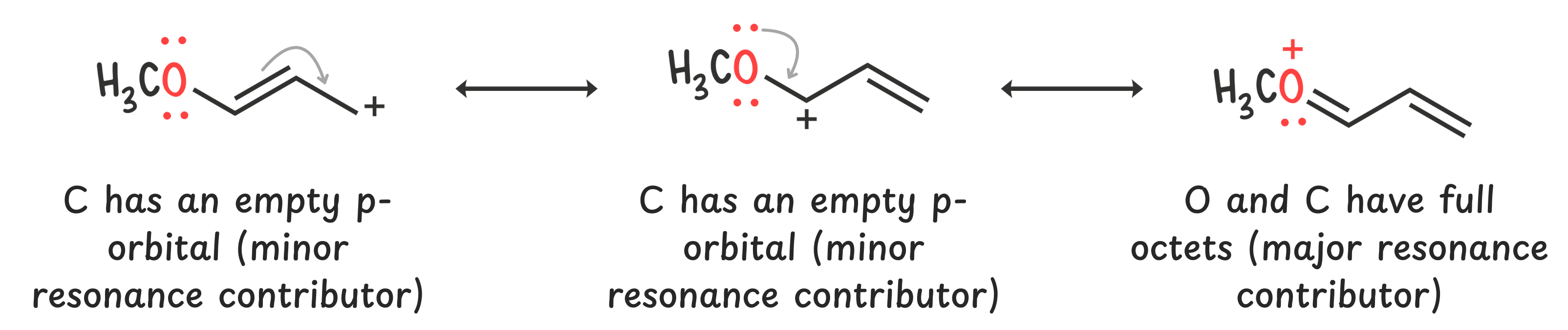
So always check for nearby heteroatoms like oxygen or nitrogen—they can offer major stabilization through lone pair donation.
Step 3: Consider factors that destabilize carbocations
Not all structural features are helpful. Some actually make carbocations less stable. Here’s what to watch for:
1️⃣ Electron-withdrawing groups
EWGs do the opposite of alkyl groups—they pull electron density away from the carbocation center via inductive effects, increasing its positive character and making it less stable.
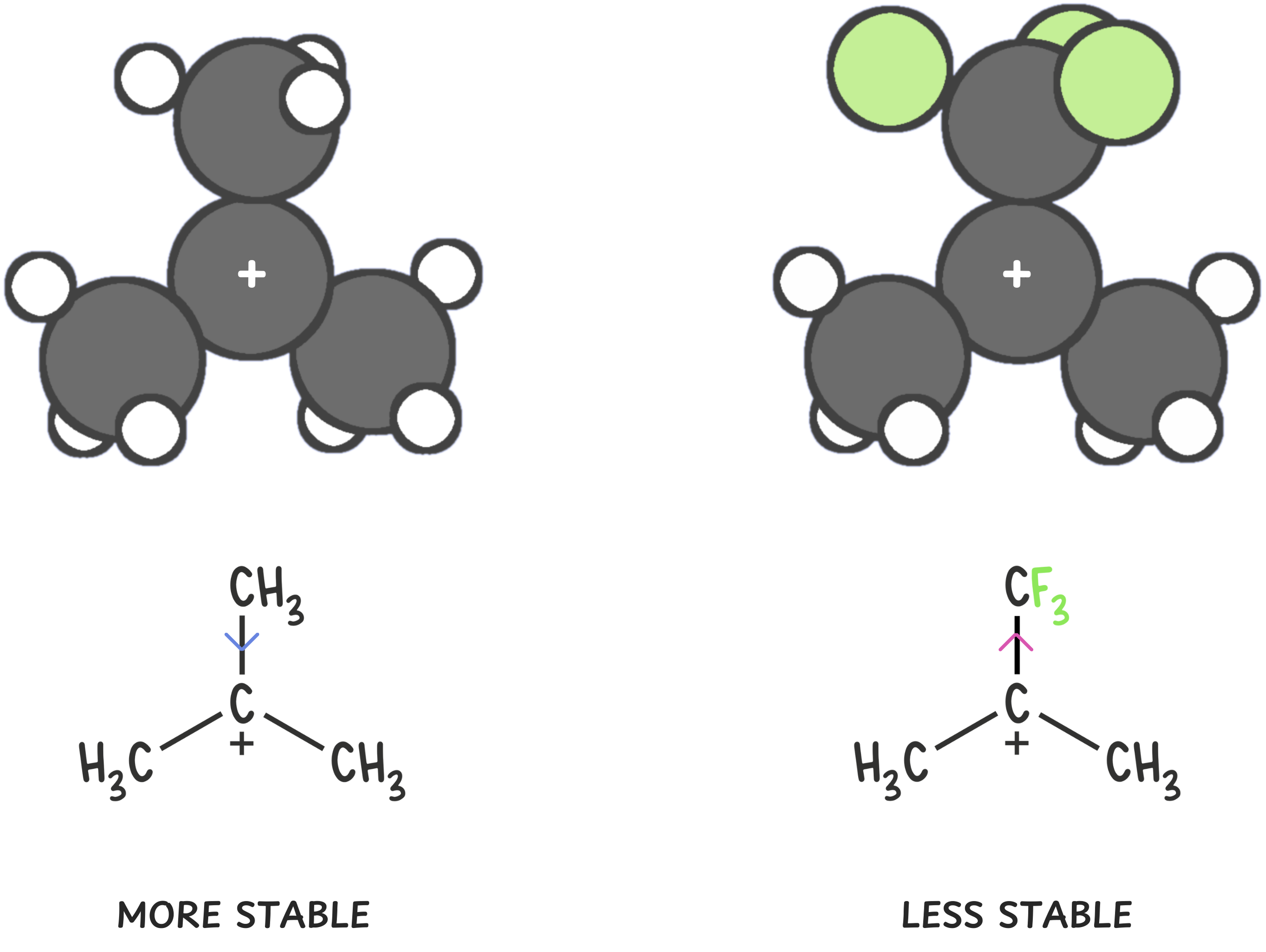
Examples include: -NO2, -CN, -CF3, carbonyls.
2️⃣ Hybridization effects
The more s-character an orbital has, the closer it is to the positively charged nucleus.
What does this mean for carbocations?
A positive charge is less stable on a carbon with high s-character.
- sp³ hybridized carbon (25% s-character) – fine for carbocations
- sp² hybridized carbon (33%) – less favorable
- sp hybridized carbon (50%) – very unlikely
So, carbocations rarely form on alkynes (sp) or alkene (sp²) centers—those orbitals are just too close to the nucleus to tolerate a positive charge.
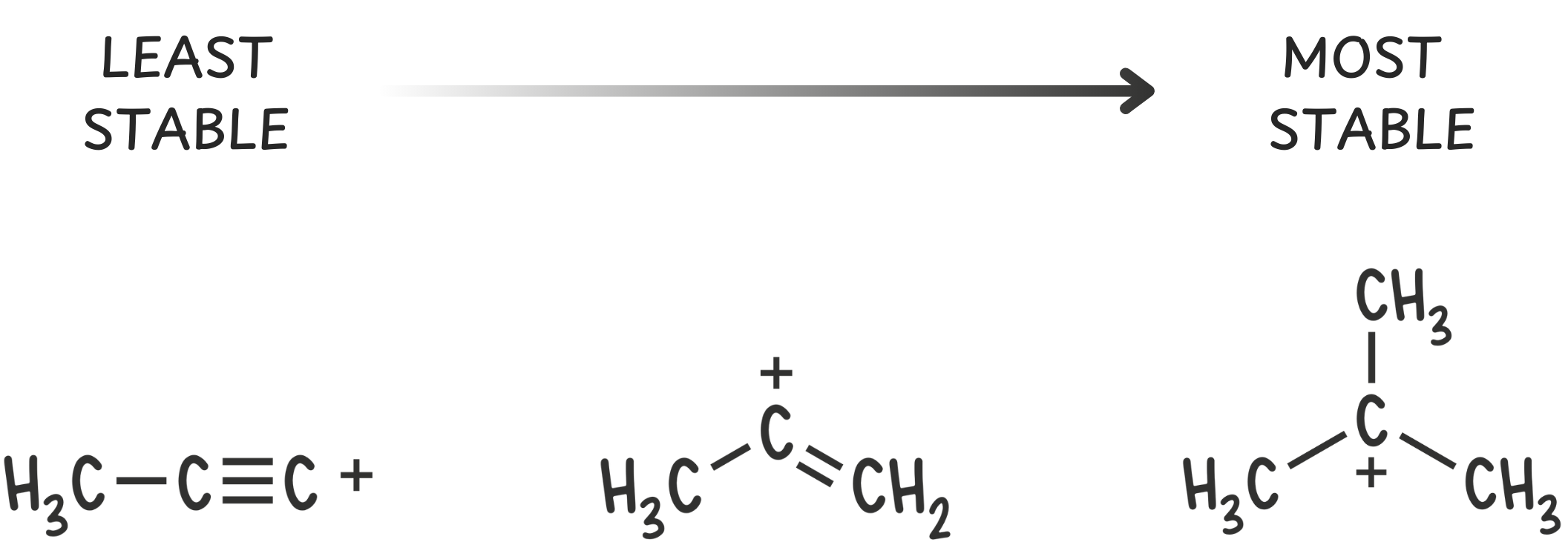
Step 4: Rank the carbocations
Now you’re ready to put it all together.
When comparing carbocations, ask:
1️⃣ What type is it? (methyl, 1°, 2°, 3°, allylic, benzylic)
2️⃣ What stabilizing features are present?
- Alkyl groups
- Resonance (π bonds or lone pairs)
3️⃣ What destabilizing features are nearby?
- Electron-withdrawing groups?
- High s-character carbon?
Once you’ve considered all of that, you can confidently rank them by stability—a critical skill in predicting reaction pathways like SN1, E1, and rearrangement mechanisms.
Got questions or thoughts? Share them below!